US Pharm. 2020;45(4):21-28.
ABSTRACT: U.S. travelers visiting or working in other parts of the world must protect themselves against viruses that can be transmitted by animals or humans. These viruses include Japanese encephalitis, typhoid fever, yellow fever, polio, rabies, and hepatitis A. Reviewing these vaccine-preventable diseases will enhance the pharmacist’s knowledge about the appropriate preventive measures to undertake in order to avoid illness. Preventive measures include basic handwashing, proper selection of clothing for activities in risk-prone areas, and vaccines. Pharmacists are on the front line to provide patient counseling and help travelers determine which vaccines are necessary to prevent diseases.
The recent global outbreak of the 2019 novel coronavirus SARS-CoV-2 (known as coronavirus disease 2019 [COVID-19]) caused the World Health Organization (WHO) to declare a global emergency. At present, no vaccine is available that can protect against COVID-19; however, vaccine-development efforts are underway. Awareness of this pandemic should alert individuals to protect themselves from vaccine-preventable diseases while traveling.
According to the CDC, travelers are advised to protect themselves with vaccines at least 4 to 6 weeks prior to traveling. This allows time for the individual to build up immunity to the vaccine. The CDC’s Travelers’ Health website contains comprehensive information and allows users to input a specific travel destination to determine recommended vaccines and precautions for that area.1 The Advisory Committee on Immunization Practices (ACIP) provides recommendations and guidelines regarding vaccines used to prevent the spread of diseases in the United States.2
Japanese Encephalitis
Overview: Japanese encephalitis virus (JEV), which is found in Asia and the western Pacific, is in the genus Flavivirus and is closely related to the yellow fever, West Nile, and dengue viruses.3 JEV is spread by mosquitoes, not by human-to-human contact.3,4 The incidence risk is low but can vary depending on the season, activities, and duration of travel.5
Symptoms: Initial presenting symptoms may consist of mild fever or a headache, followed by progression to severe disease. Symptoms of progression include rapid onset of high fever, disorientation, headache, neck stiffness, coma, seizures, and spastic paralysis, and death may ensue. Gastrointestinal pain and vomiting may be the initial symptoms in children. The incubation period is 4 to 14 days. Persons with disease symptoms have a case-fatality rate of 20% to 30%. Patients who survive JEV experience lifelong repercussions, such as paralysis, inability to speak, recurrent seizures, and other intellectual or neurologic consequences.3-5
Diagnosis: To rule out other causes of encephalitis, the WHO recommends testing for JEV-specific immunoglobulin M (IgM) antibody using a cerebrospinal fluid (CSF) or serum sample. CSF sampling reduces false-positive rates from a previous infection or vaccination.4
Prevention: Currently, there is no antiviral treatment for JEV, and patients are managed with symptomatic care.4 The ACIP recommends the JE vaccine (Ixiaro) for travelers spending 1 month or longer in endemic areas, including rural agricultural areas of Asia and areas containing a large number of vector mosquitoes when production and flooding irrigation occur. The vaccine is also recommended for laboratory workers who are at risk for exposure to JEV. The ACIP does not recommend routine vaccination for travelers visiting urban areas or those whose visit is shorter than 1 month unless the traveler plans activities that will increase the risk of exposure, such as camping or hiking.5 See TABLE 1 for JE vaccine dosing and special considerations.6
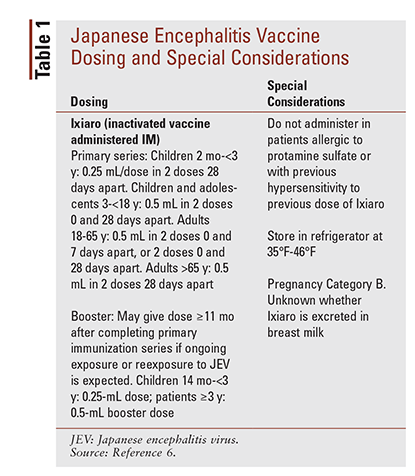
Adverse Effects: Some adverse effects of Ixiaro are local and systemic adverse events, headache, flulike illness, fatigue, and myalgia. In children, common side effects are febrile seizures and pneumonia.6
Typhoid Fever
Overview: Typhoid fever is prevalent in many parts of the Caribbean, East and Southeast Asia, Africa, and Central and South America but is uncommon in the U.S. Typhoid fever and paratyphoid fever are caused by Salmonella serotypes typhi and paratyphi, respectively.7 The bacteria can spread when the feces of an infected person contaminates food or water and when food is prepared by someone who does not wash the hands before handling food.8 When a contaminated food or beverage is ingested, the bacteria can multiply and spread into the bloodstream, resulting in typhoid fever.9
Symptoms: Typhoid symptoms include fatigue, weakness, loss of appetite, long-lasting high fever, rash (flat, pink spots), stomach pain, diarrhea, constipation, or cough. People can unknowingly spread typhoid to others even when their symptoms are gone.8,9
Diagnosis: Blood culture is the primary method for diagnosing typhoid. Stool, urine, or bone marrow samples may also be used for the diagnosis of typhoid or paratyphoid. The most sensitive test for Salmonella typhi is bone marrow culture.10
Prevention: Typhoid vaccine can prevent typhoid fever, which may be life-threatening. Fever can last for weeks to months if antibiotics are not taken. Approximately 30% of people who are not treated die from complications of typhoid fever. Fewer antibiotic treatment options are available because drug-resistant typhoid bacteria have become more common in many parts of the world.11
Patients can carry Salmonella typhi or paratyphi even if symptoms are not present. Bacteria can be spread to others and the illness could return, so a physician must perform a diagnostic workup to determine whether the patient can return to work or school. To lessen the chance of spreading bacteria to others, patients should finish their recommended course of antibiotic therapy, wash their hands with soap and water after using the bathroom, and avoid preparing and serving food to others.11
Typhoid vaccine (Vivotif, Typhim Vi) is recommended for persons at high risk for exposure to typhoid bacteria, such as laboratory workers, those in close contact with another infected person, and those traveling to Africa, Asia, or Central and South America.12 See TABLE 2 for typhoid vaccine dosing recommendations and special considerations.13,14

Adverse Effects: Adverse effects of Typhim Vi, including pain, headache, fever, and redness or swelling at the injection site, are usually mild and resolve in a few days. Common adverse effects from Vivotif include fever or headache; less common side effects are vomiting, stomach pain, and rash. It is rare to experience serious adverse effects from either vaccine.8
Yellow Fever
Overview: Yellow fever, which is caused by RNA viruses in the genus Flavivirus, is related to JEV, West Nile virus, and St. Louis encephalitis. Yellow fever virus is transmitted to human and nonhuman primates by animals, chiefly daytime-biting female Aedes aegypti mosquitoes and mosquitoes in the Haemagogus genus.15 Precautions against yellow fever virus should be taken in persons traveling to Africa and South America.16
Symptoms: Most patients infected with yellow fever are asymptomatic or have some mild symptoms. The incubation period occurs approximately 3 to 6 days before symptoms manifest. Initial symptoms, which include body aches, chills, severe headaches, nausea, vomiting, sudden onset of fever, back pain, weakness, and fatigue, generally last for about 1 week, but sometimes fatigue and weakness linger for months. One in seven people who develop initial symptoms experience a brief remission lasting a few hours or a day before progressing to more severe symptoms, including high fever, bleeding, shock, jaundice, multiorgan failure, and death. Approximately 30% to 60% of patients who develop severe symptoms will die.15,17
Diagnosis: To diagnose yellow fever, a serum sample is analyzed in the laboratory. Virus-specific IgM and neutralizing antibodies are found in the serum of patients infected with yellow fever. The healthcare provider also collects information about the dates and locations where the patient traveled, activities engaged in while traveling, and clinical features in order to make a diagnosis.18
Prevention: Yellow fever vaccine (YF-VAX) is a live, weakened form of the virus administered as a single injection.17 Infected persons are likely immune from future infection. The ACIP currently does not require booster vaccines, owing to sustained immunity from a single vaccination.19 Travelers should consult the CDC website to determine whether YF-VAX is required.1 Some countries require a Certificate of Vaccination before allowing entry into the country.20 The traveler should be issued a waiver if the vaccination is contraindicated for any medical reason, but it is up to the destination country to determine whether entry is allowed. See TABLE 3 for YF-VAX dosing and special considerations.20,21 In addition, the following are preventive measures travelers should take to prevent mosquito bites during the day18:
• Spray or presoak outer layer of clothing and gear with permethrin.
• Wear long-sleeved, breathable, neutral-colored clothing.
• Apply sunscreen, wait 20 minutes, and then apply bug repellent.
• Apply 20%-30% DEET or 20% picaridin to exposed skin and follow the manufacturer’s reapplication directions.
• Remove open containers of water around living spaces and use screens on windows and doors.
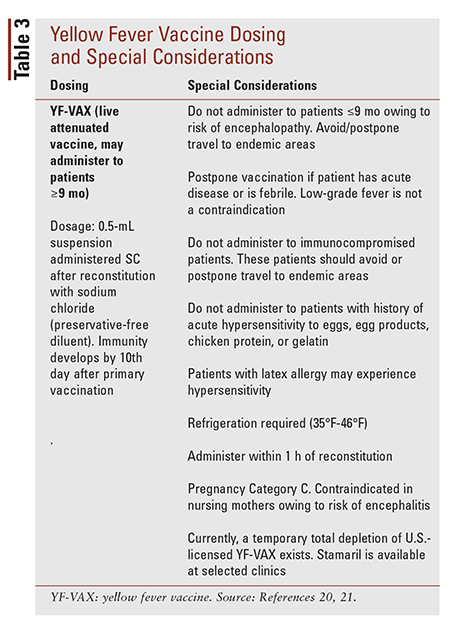
Adverse Effects: Common adverse effects of YF-VAX are headache, fever, swelling and pain at the injection site, and muscle aches or discomfort. Severe adverse effects are rare, but YF-VAX has been associated with multiple organ failure and brain inflammation.20
Availability: Currently there is a national shortage of YV-VAX. Sanofi Pasteur, the manufacturer of YV-VAX, is experiencing production delays due to transitioning to a new facility. The FDA has accepted an expanded-access investigational New Drug Application to allow the importation of Stamaril, a vaccine produced in France by Sanofi Pasteur, for use in the meantime. Stamaril, which is used in more than 70 countries, is made with the same vaccine substrain as YV-VAX and is currently being allocated for use at certain clinics in the U.S.22
Polio
Overview: Polio (poliomyelitis) is an infectious viral disease that affects the central nervous system and can cause temporary or permanent paralysis. Polio is caused by poliovirus types 1, 2, and 3. Poliovirus is transmitted through person-to-person contact; a person can get infected by contaminated water or food, for example, and then transmit the virus to others via coughing and sneezing. Infected individuals are contagious before developing symptoms and up to 2 weeks after symptoms resolve. Poliovirus can live in fecal matter for several weeks, allowing the virus to be easily transmitted in regions with poor sanitation.23 Polio occurs in Afghanistan, Nigeria, and Pakistan, and poliovirus transmission in these countries has never been interrupted. Four regions are polio free: the Americas, Europe, Southeast Asia, and the Western Pacific.24
Symptoms: Most people infected with poliovirus are asymptomatic. Minor symptoms, including sore throat, headache, fever, fatigue, nausea, and stomach pain, occur in approximately 4% to 8% of infections. A few days after minor symptoms resolve, about 1% to 5% of patients develop aseptic meningitis. Serious symptoms include paralysis, paresthesia, brain or spinal problems, and permanent disability or death. Paralytic poliomyelitis occurs in one of every 1,000 infections.23,25
Diagnosis: Collection of two stool specimens 24 hours apart increases the likelihood of isolating poliovirus. The most sensitive method of diagnosing poliovirus infection is to isolate the virus in culture.26
Prevention: Vaccination against poliovirus (IPOL) is recommended for individuals traveling to endemic countries, healthcare workers caring for patients with poliovirus, and laboratory employees who handle specimens containing poliovirus. See TABLE 4 for dosing and special considerations for the polio vaccine.23

Adverse Effects: Common adverse effects of IPOL include swelling, injection-site pain, fatigue, fever, loss of appetite, vomiting, and irritability.23
Rabies
Overview: The saliva of infected mammals can transmit the rabies virus to humans who are bitten or scratched. Rabies virus can be fatal because of encephalomyelitis. The incubation period can last from 5 days to several years; however, it is usually between 20 and 60 days. In the U.S., rabies is uncommon in humans. Sources of human rabies infection in the U.S. include skunks, foxes, raccoons, and bats; in other countries, unvaccinated dogs are the cause.27
Symptoms: Rabies symptoms begin with prodromal symptoms, including fatigue, headache, malaise, anorexia, fever, pain, and paresthesia. As the disease progresses, patients may have hallucinations, seizures, anxiety, agitation, and irritability. In the advanced stage, patients may experience aerophobia, hydrophobia, hypersalivation, hyperactivity, disorientation, seizures, and eventually paralysis, coma, and death.27
Diagnosis: To confirm that rabies virus has been transmitted by an infected animal to a human, several tests are required. Samples of saliva, spinal fluid, and serum and skin biopsies of hair follicles at the nape of the neck may be used.28
Prevention: There are several ways to prevent rabies infection, such as avoiding wildlife, vaccinating pets, and seeking medical attention before symptoms begin.29 It is recommended that the following high-risk groups obtain preexposure rabies vaccination as a preventive: spelunkers, veterinarians and veterinary students, animal handlers, and laboratory workers handling rabies live vaccine and immune globulin. Additionally, vaccination should be considered for international travelers who may have limited access to immediate care and who may come in contact with animals that have rabies. If an individual is bitten or scratched, it is important to cleanse the wounded area with soap and water, followed by irrigation of the wound with a virucidal agent such as povidone-iodine solution.30
There are two formulations of rabies vaccines: human diploid cell vaccine (Imovax) and purified chick embryo cell vaccine (RavAvert).31 Four formulations of rabies immune globulin are available: Imogam Rabies-HT, Kedrab, HyperRAB, and HyperRAB S/D.32 HyperRAB (300 IU/mL) is twice the concentration of HyperRAB S/D (150 IU/mL), which is a solvent/detergent-treated product. Patients receiving HyperRAB (300 IU/mL) are getting a lower volume per dose since it is a higher concentration.33
See TABLE 5 for dosing and special considerations for rabies vaccines.31,32 Preexposure vaccination is recommended to provide protection to those at high risk for exposure. It also helps protect patients who may not have immediate access to care due to delays if exposed to the rabies virus. Preexposure vaccination also eliminates the need for immune globulin, thereby reducing the number of injections.27

Adverse Effects: Common adverse effects of rabies vaccines include injection-site rash, abdominal pain, nausea, localized enlarged lymph nodes, arthralgia, myalgia, dizziness, flulike symptoms, malaise, and headache. Serious adverse effects are anaphylaxis, encephalitis, Guillain-Barré syndrome, and transfusion reaction due to serum protein reaction.31
Hepatitis A
Overview: Hepatitis A virus (HAV) is transmitted person-to-person through the fecal-oral route by consumption of contaminated food or water. Those at risk for contracting HAV include travelers to countries with high or intermediate endemic areas, men who have sex with men, and users of injectable drugs, and persons working with nonhuman primates; those who come in close contact with an infected person may be susceptible to the virus.34,35
Diagnosis: To diagnose HAV, specific IgG antibodies must be detected in the blood.36
Symptoms: Adults present with the following symptoms: fatigue, nausea, jaundice, low appetite, and stomach pain. Children aged younger than 6 years usually do not show symptoms or have an infection that is not recognized. HAV is usually self-limiting and does not advance to chronic infection. There is no treatment for HAV other than supportive care such as rest, balanced nutrition, and fluids.34
Prevention: The best way to prevent HAV infection is vaccination (Vaqta, Havrix), which should occur at least 2 weeks before anticipated exposure to HAV. Antibodies produced in response to HAV infection confer lifelong protection against reinfection. See TABLE 6 for dosing and special considerations for hepatitis A vaccines.35
Adverse Effects: Common adverse effects of hepatitis A vaccines are pain, injection-site swelling or erythema, loss of appetite, nausea, irritability, headache, somnolence, fatigue, fever, and malaise. Serious adverse effects include thrombocytopenia, anaphylactoid reaction, seizure, angioedema, and syncope accompanied by neurologic signs such as visual disturbance, tonic-clonic limb movements, and paresthesia.34,35
Conclusion
Before traveling, it is important for patients to review which vaccines pertain to their destination. The CDC website is a comprehensive knowledge base for patients and healthcare providers. Pharmacists should counsel patients about travel vaccines and their adverse effects, as well as provide information about nonpharmacologic practices to lessen the transmission risk.
REFERENCES
1. CDC. Travelers’ health. wwwnc.cdc.gov/travel. Accessed January 15, 2020.
2. CDC. ACIP recommendations. www.cdc.gov/vaccines/acip/recommendations.html. Accessed January 15, 2020.
3. FDA. Ixiaro. www.fda.gov/vaccines-blood-biologics/vaccines/ixiaro. Accessed January 15, 2020.
4. World Health Organization. Japanese encephalitis. www.who.int/news-room/fact-sheets/detail/japanese-encephalitis. Accessed January 15, 2020.
5. Hills SL, Lindsey NP, Fischer M. Japanese encephalitis. 2020 Yellow Book. wwwnc.cdc.gov/travel/yellowbook/2020/travel-related-infectious-diseases/japanese-encephalitis. Accessed January 15, 2020.
6. Ixiaro (Japanese encephalitis vaccine) package insert. Gaithersburg, MD: Valneva USA, Inc; September 2018.
7. CDC. Typhoid fever and paratyphoid fever. www.cdc.gov/typhoid-fever/index.html. Accessed January 15, 2020.
8. Vaccines.gov. Typhoid fever. www.vaccines.gov/diseases/typhoid_fever. Accessed January 15, 2020.
9. CDC. Typhoid fever and paratyphoid fever: questions and answers. www.cdc.gov/typhoid-fever/sources.html. Accessed January 15, 2020.
10. Mayo Clinic. Typhoid fever. www.mayoclinic.org/diseases-conditions/typhoid-fever/diagnosis-treatment/drc-20378665. Accessed January 15, 2020.
11. CDC. Typhoid fever and paratyphoid fever: symptoms and treatment. www.cdc.gov/typhoid-fever/symptoms.html. Accessed January 15, 2020.
12. CDC. Typhoid vaccine information statements. www.cdc.gov/vaccines/hcp/vis/vis-statements/typhoid.html. Accessed January 15, 2020.
13. Typhim Vi (typhoid Vi polysaccharide vaccine) package insert. Swiftwater, PA: Sanofi Pasteur, Inc; March 2014.
14. Vivotif (typhoid vaccine live oral Ty21a) package insert. Miami Lakes, FL: Crucell Vaccines Inc; September 2013.
15. International Association for Medical Assistance to Travellers. Yellow fever. www.iamat.org/risks/yellow-fever. Accessed January 15, 2020.
16. CDC. Transmission of yellow fever virus. www.cdc.gov/yellowfever/transmission/index.html. Accessed January 15, 2020.
17. CDC. Yellow fever vaccine. www.cdc.gov/yellowfever/vaccine. Accessed January 15, 2020.
18. CDC. Yellow fever: diagnostic testing. www.cdc.gov/yellowfever/healthcareproviders/healthcareproviders-diagnostic.html. Accessed January 15, 2020.
19. Staples JE, Bocchini JA Jr, Rubin L, Fischer M. Yellow fever vaccine booster doses: recommendations of the Advisory Committee on Immunization Practices, 2015. MMWR Morb Mortal Wkly Rep. 2015;64(23);647-650.
20. YF-VAX (yellow fever vaccine) package insert. Swiftwater, PA: Sanofi Pasteur, Inc; May 2013.
21. CDC. Clinical update announcement: temporary total depletion of US licensed yellow fever vaccine addressed by availability of Stamaril vaccine at selected clinics. wwwnc.cdc.gov/travel/news-announcements/yellow-fever-vaccine-access. Accessed January 15, 2020.
22. Sanofi. Yellow fever vaccine information. www.sanofi.us/en/products-and-resources/vaccines/yellow-fever-vaccine-information. Accessed January 15, 2020.
23. IPOL (poliovirus vaccine inactivated) package insert. Swiftwater, PA: Sanofi Pasteur, Inc; August 2015.
24. CDC. Our progress against polio. www.cdc.gov/polio/progress/index.htm. Accessed January 15, 2020.
25. Passport Health. What is polio? www.passporthealthusa.com/vaccinations/polio. Accessed January 15, 2020.
26. CDC. Polio: diagnostic methods. www.cdc.gov/polio/what-is-polio/lab-testing/diagnostic.html. Accessed January 15, 2020.
27. RavAvert (rabies vaccine) package insert. Research Triangle Park, NC: GlaxoSmithKline; September 2019.
28. CDC. Rabies: diagnosis in animals and humans. www.cdc.gov/rabies/diagnosis/animals-humans.html. Accessed January 15, 2020.
29. CDC. Vaccine information statements: rabies VIS. www.cdc.gov/vaccines/hcp/vis/vis-statements/rabies.html. Accessed January 15, 2020.
30. Imovax (rabies vaccine) package insert. www.vaccineshoppe.com/image.cfm?doc_id=5983&image_type=product_pdf. Accessed January 15, 2020.
31. Micromedex. Drug summary for rabies vaccine. Greenwood Village, CO: Truven Health Analytics; 2020. www.micromedexsolutions.com. Accessed January 15, 2020.
32. Micromedex. Drug summary for rabies immune globulin. Greenwood Village, CO: Truven Health Analytics; 2020. www.micromedexsolutions.com. Accessed January 15, 2020.
33. Hypermunes. HyperRAB: frequently asked questions. www.hypermunes.com/en/hcp/faq. Accessed January 15, 2020.|
34. World Health Organization. Hepatitis A: key facts. www.who.int/news-room/fact-sheets/detail/hepatitis-a. Accessed January 15, 2020.
35. CDC. Hepatitis A questions and answers for health professionals. www.cdc.gov/hepatitis/hav/havfaq.htm#travel. Accessed February 14, 2020.
36. Havrix (hepatitis A vaccine) package insert. Research Triangle Park, NC: GlaxoSmithKline; December 2018.
37. Vaqta (hepatitis A vaccine) package insert. Whitehouse Station, NJ: Merck & Co, Inc; November 2019.
To comment on this article, contact rdavidson@uspharmacist.com.





