US Pharm. 2022;47(2):HS9-HS12.
ABSTRACT: Many patients diagnosed with pericarditis are hospitalized for acute or recurrent episodes. Hospitalizations for pericarditis are associated with an increased risk of morbidity and mortality, and many patients experience poor quality of life. Although the 2015 European Society of Cardiology guidelines provide recommendations on the management of patients with pericarditis, some of the dosage forms discussed in the guidelines are not available in the United States; moreover, there have been significant updates in the management of hospitalized patients with pericarditis. Inpatient pharmacists can play a vital role in the management of patients with pericarditis regardless of whether they serve in a centralized or decentralized capacity.
Pericarditis is the most common form of pericardial disease.1 In the Western Hemisphere, pericarditis most often has idiopathic, viral, or injurious etiologies.1 Patients with viral or idiopathic etiologies have a particularly high risk of recurrence after the first acute pericarditis (AP) index event, and an even higher risk of recurrence with multiple recurrences of pericarditis.2-6 Hospitalized patients diagnosed with idiopathic AP are at increased risk for adverse outcomes, such as relatively long length of hospital stay, use of procedures for managing pericarditis, healthcare burden, and mortality.7 Additionally, disparities of care are evident among hospitalized patients diagnosed with idiopathic AP, including women, certain minorities, and payor type.7
Guidance documents on the care of hospitalized patients with pericarditis are available, including the 2015 European Society of Cardiology (ESC) guidelines for diagnosis and management of pericardial diseases.1,8 However, since the publication of the 2015 ESC guidelines, there have been significant updates in the management of patients diagnosed with pericarditis in the hospital.9 In the majority of cases, pharmacotherapy is the mainstay of treatment in hospitalized patients diagnosed with idiopathic, viral, or injurious etiologies of pericarditis; however, these pharmacotherapies are associated with significant drug-drug/disease interactions and adverse effects (AEs).1 Hospital or inpatient pharmacists are well positioned to participate in and impact the care of patients diagnosed with pericarditis.10 This article describes the contemporary management of hospitalized patients diagnosed with pericarditis, including the role of the pharmacist.
Diagnosis and Evaluation
Given that one of the most common symptoms of pericarditis is chest pain, patients typically are initially evaluated in the emergency department and are often admitted to internal medicine, general cardiology, or cardiac intensive care services to rule out other differential diagnoses of chest pain (e.g., acute coronary syndromes, aortic dissection, or dysrhythmias).1 Pericarditis is usually diagnosed using at least two of the following criteria: pericarditic chest pain (pleuritic chest pain that may be positional [i.e., worsens upon lying supine and improves upon leaning forward] and may radiate to the trapezial ridges); new or worsening pericardial effusion noted on echocardiography; pericardial friction rub upon auscultation; and classic ECG signs (i.e., PR-segment depression and/or ST-segment elevation).1 Cardiac MRI and cardiac computed tomography are also useful for diagnosing pericarditis.1 Inflammatory markers (e.g., elevated C-reactive protein [CRP]) may be useful not only for diagnosis of pericarditis, but also for prognosis and response to treatment.1,11
Management
Pharmacotherapy for the treatment of idiopathic pericarditis includes aspirin (ASA)/nonsteroidal anti-inflammatory drugs (NSAIDs), colchicine, glucocorticoids (GCs), and immunotherapies (TABLE 1).
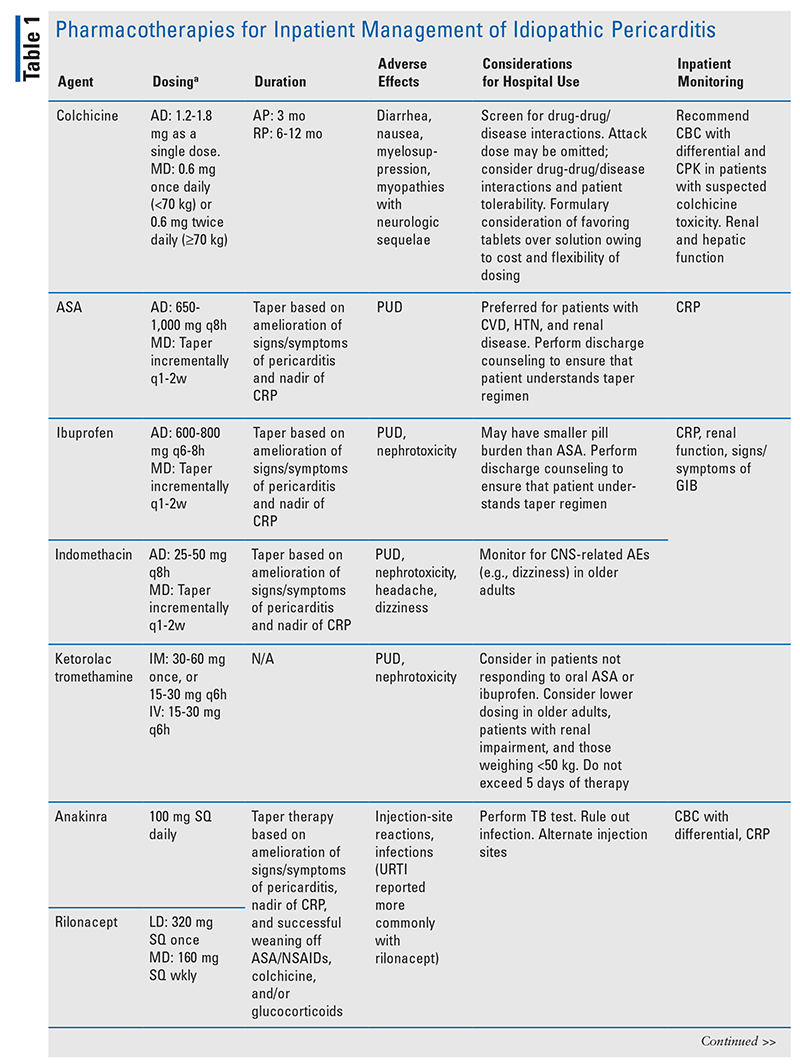
ASA/NSAIDs: High doses of ASA and NSAIDs are recommended for management of pericarditic chest pain in hospitalized patients with pericarditis.1 ASA and NSAIDs do not impact disease progression associated with pericarditis. However, because failure to respond to NSAIDs within a week of initiation is associated with a poor prognosis, the appropriate use of relatively high doses of ASA/NSAIDs is of paramount importance to patient success.9,12 NSAIDs such as ibuprofen, indomethacin, and ketorolac tromethamine have been most studied in patients with pericarditis.8 The use of parenteral NSAIDs such as ketorolac is recommended in patients who do not respond to oral ASA or NSAIDs.1 High doses should be used for 1 to 2 weeks, and tapering should be initiated upon return of normal CRP concentration and amelioration of any signs or symptoms of pericarditis.1,8,9,11 The dosing and tapering regimens should consider other drugs, concomitant disease states, affordability, risk of AEs (e.g., dyspepsia, bleeding, dizziness), and patient age. Patients should receive gastroprotection with a proton pump inhibitor during ASA/NSAID therapy. Deprescribing of gastroprotection should take place upon discontinuation of ASA/NSAID therapy.1
Colchicine: Colchicine, as first-line treatment, should be part of adjuvant ASA/NSAID therapy.1 This agent has been shown to reduce the risk of recurrence in idiopathic AP and prevent future recurrences in patients with idiopathic recurrent pericarditis (RP).2-6 It may also lessen time to remission and hasten time to pain control.2-6 Colchicine dosing is weight-based and is also guided by drug-drug/disease interactions and propensity for AEs.2-6,9,13 The most common AEs of colchicine are gastrointestinal, chiefly diarrhea.13 Potentially life-threatening AEs include myelosuppression and neuromyopathies.9,13 An attack dose (1-2 mg) was administered in some of the landmark studies, but it may be omitted if the clinician is concerned about an increased risk of AEs.1-6,8,9 Maintenance dosing (0.5-1 mg) consists of daily or twice-daily dosing based on patient tolerability for 3 months for idiopathic AP, and 6 to 12 months for idiopathic RP.2-6,9 The dosage form used in the landmark trials (i.e., 0.5-mg tablet) is not available in the United States. Several formulations (all of them 0.6 mg) are available in the U.S., including scored tablet, capsule, and oral solution. Given colchicine’s narrow therapeutic index, dosing should be carefully extrapolated using U.S. formulations.9,13
Pharmacists should initiate colchicine in the hospital in order to ensure appropriate pharmacovigilance for AEs and drug-drug/disease interactions.9,13 Colchicine is metabolized by the liver via CYP3A4 and eliminated renally, along with elimination via P-glycoprotein (P-gp).12 Therefore, inpatient pharmacists should screen for drug-drug interactions.9,10 Depending on the dosage form, colchicine may be contraindicated in certain patients who present with drug-drug and drug-disease interactions.9,10 For example, the scored-tablet formulation is contraindicated in patients receiving a strong CYP3A4 inhibitor or P-gp inhibitor in the presence of renal or hepatic impairment, whereas the oral-solution and capsule formulations are contraindicated in patients receiving a strong CYP3A4 inhibitor or P-gp inhibitor in the presence of both renal and hepatic impairment.9,10
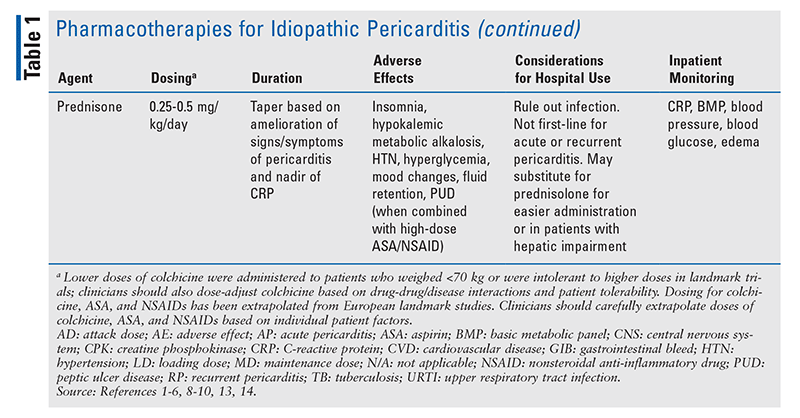
GCs: Although historically they have been used to treat pericarditis, GCs are not recommended first-line owing to increased risk of recurrence, dependency, and both short-term and long-term AEs (including peptic ulcer disease when combined with NSAIDs, hyperglycemia, electrolyte disturbances, mood disorders, osteoporosis, immunosuppression, hypertension, Cushing syndrome, fluid retention, weight gain, and insomnia).1,8,9 GCs should be reserved for patients who fail ASA/NSAID plus colchicine therapy, have intolerances or contraindications to ASA/NSAID or colchicine therapy, or have indications for GCs (e.g., certain autoimmune diseases).1 GCs should be initiated at lower doses (i.e., 0.25-0.5 mg/kg) and weaned slowly based on the dose, and when CRP has normalized and any signs or symptoms of pericarditis have ameliorated.1,9,14
Immunotherapies: Many patients with RP may experience multiple recurrences and significant disease-state burden, necessitating combination therapy with ASA/NSAIDs, colchicine, and GCs.1,8 Given many of the aforementioned pharmacotherapies’ propensity for AEs and considering the need for steroid-sparing regimens, immunotherapeutic agents targeting immune system–mediated inflammation pathways have been investigated for the management of RP.8,9 The interleukin (IL)-1 antagonists anakinra (IL-1 receptor antagonist) and rilonacept (IL-1 trap) have the most robust data of the immunotherapeutic agents used to treat RP.8,9,15 More data exist on the use of anakinra in patients with RP.8,9 However, rilonacept recently received orphan-drug FDA approval for the prevention and treatment of adults and children with RP and elevated CRP.15 Both of these IL antagonists are associated with local injection-site reactions, and they should not be used in immunocompromised patients.8,15 Rilonacept is also associated with upper respiratory tract infections and hyperlipidemia.15 Other immunotherapies associated with improved outcomes in patients with RP—albeit with less robust data—include parenteral immunoglobulin, azathioprine, and hydroxychloroquine.16-18
Role of the Inpatient Pharmacist
Inpatient pharmacists can play a vital role in the management of patients with pericarditis regardless of whether they serve in a centralized or decentralized capacity.10 At the point of direct patient care, inpatient pharmacists can help prescribers design pharmacotherapy regimens based on landmark studies and the 2015 ESC guidelines, including ensuring the use of first-line medications proven to decrease risk of recurrence and manage symptoms. Given the complexity of the data regarding which patients are candidates for immunotherapy, pharmacists are well positioned to interpret clinical trials supporting appropriate use of immunotherapies, especially anakinra and rilonacept. This can be done both from an administrative perspective (e.g., formulary restriction) and from the point of care.10
All of the medications used to manage pericarditis are associated with significant AEs; therefore, pharmacists are invaluable in identifying and mitigating the risk of AEs. Additionally, many patients with pericarditis present with multiple comorbidities, increasing the risk of drug-drug/disease interactions.10 Pharmacists are qualified to help identify and address such interactions, ensuring the safe use of pharmacotherapy.10 As the point of patient discharge approaches, inpatient pharmacists can facilitate the creation of tapering schedules for ASA/NSAIDs and/or GCs as well as drug acquisition and medication access, especially with more expensive medications, such as the capsule or oral-solution colchicine formulations, as well as immunotherapeutic agents.10 Upon patient discharge, pharmacists can assist in transitions of care to ensure timely follow-up and duration of pharmacotherapy as well as patient-discharge counseling.10 Disease-state education may also be considered, as many patients (e.g., athletes) may need to comply with exercise restriction for several months or until signs and symptoms of AP or RP resolve.1,10
Conclusion
Many patients diagnosed with pericarditis are hospitalized for acute or recurrent episodes. Hospitalizations for pericarditis are associated with an increased risk of morbidity and mortality, and many patients experience poor quality of life. Inpatient pharmacists can play a key role in the management of hospitalized patients with pericarditis. Specifically, the inpatient pharmacist can ensure appropriate dosing (e.g., in patients with drug-disease/drug interactions) of medications; prevent and identify AEs; ensure guideline- and landmark study–directed pharmacotherapies; provide administrative support with formulary management; and develop appropriate-use criteria for pharmacotherapies that are used to treat pericarditis in the hospital.
REFERENCES
1. Adler Y, Charron P, Imazio M, et al. 2015 ESC Guidelines for the diagnosis and management of pericardial diseases: the Task Force for the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology (ESC) endorsed by: the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2015;36:2921-2964.2. Imazio M, Bobbio M, Cecchi E, et al. Colchicine in addition to conventional therapy for acute pericarditis: results of the COlchicine for acute PEricarditis (COPE) trial. Circulation. 2005;112:2012-2016.
3. Imazio M, Brucato A, Cemin R, et al. A randomized trial of colchicine for acute pericarditis. N Engl J Med. 2013;369:1522-1528.4. Imazio M, Bobbio M, Cecchi E, et al. Colchicine as first-choice therapy for recurrent pericarditis: results of the CORE (COlchicine for REcurrent pericarditis) trial. Arch Intern Med. 2005;165:1987-1991.
5. Imazio M, Brucato A, Cemin R, et al. Colchicine for recurrent pericarditis (CORP): a randomized trial. Ann Intern Med. 2011;155:409-414.6. Imazio M, Belli R, Brucato A, et al. Efficacy and safety of colchicine for treatment of multiple recurrences of pericarditis (CORP-2): a multicentre, double-blind, placebo-controlled, randomised trial. Lancet. 2014;383:2232-2237.
7. Schwier NC, Tran NT, Skrepnek GH. Clinical and economic outcomes associated with hospitalizations for acute idiopathic pericarditis in the United States. Heart Lung. 2021;50:825-831.8. Chiabrando JG, Bonaventura A, Vecchié A, et al. Management of acute and recurrent pericarditis: JACC state-of-the-art review. J Am Coll Cardiol. 2020;75:76-92.
9. Schwier NC, Tsui J, Perrine JA, et al. Current pharmacotherapy management of children and adults with pericarditis: prospectus for improved outcomes. Pharmacotherapy. 2021;41:1041-1055.10. Schwier NC. Pharmacists’ role in the management of acute and recurrent pericarditis: inpatient and outpatient perspectives. J Pharm Pract. 2020;33:838-845.
11. Imazio M, Brucato A, Maestroni S, et al. Prevalence of C-reactive protein elevation and time course of normalization in acute pericarditis: implications for the diagnosis, therapy, and prognosis of pericarditis. Circulation. 2011;123:1092-1097.12. Imazio M, Cecchi E, Demichelis B, et al. Indicators of poor prognosis of acute pericarditis. Circulation. 2007;115:2739-2744.
13. Schwier NC. Pharmacotherapeutic considerations for using colchicine to treat idiopathic pericarditis in the USA. Am J Cardiovasc Drugs. 2015;15:295-306.14. Imazio M, Brucato A, Cumetti D, et al. Corticosteroids for recurrent pericarditis: high versus low doses: a nonrandomized observation. Circulation. 2008;118:667-671.
15. Klein AL, Imazio M, Cremer P, et al. Phase 3 trial of interleukin-1 trap rilonacept in recurrent pericarditis. N Engl J Med. 2021;384:31-41.16. Imazio M, Lazaros G, Picardi E, et al. Intravenous human immunoglobulins for refractory recurrent pericarditis: a systematic review of all published cases. J Cardiovasc Med (Hagerstown). 2016;17:263-269.
17. Vianello F, Cinetto F, Cavraro M, et al. Azathioprine in isolated recurrent pericarditis: a single centre experience. Int J Cardiol. 2011;147:477-478.18. Lazaros G, Antonopoulos AS, Antonatou K, et al. Hydroxychloroquine for colchicine-resistant glucocorticoid-dependent idiopathic recurrent pericarditis: a pilot observational prospective study. Int J Cardiol. 2020;311:77-82.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
To comment on this article, contact rdavidson@uspharmacist.com.





