US Pharm.
2007;1:HS-3-HS-17.
Tremendous
advancements have been made in our understanding of immunology and
transplantation in the last 20 years. Kidney transplant patients now benefit
from modern immunosuppressive strategies and agents that have greatly reduced
the chance of acute cellular rejection of the transplanted organ. Although the
incidence of acute cellular rejection is much lower due to better matching
techniques and better drugs, long-term kidney survival rates are still
marginal at best.1,2
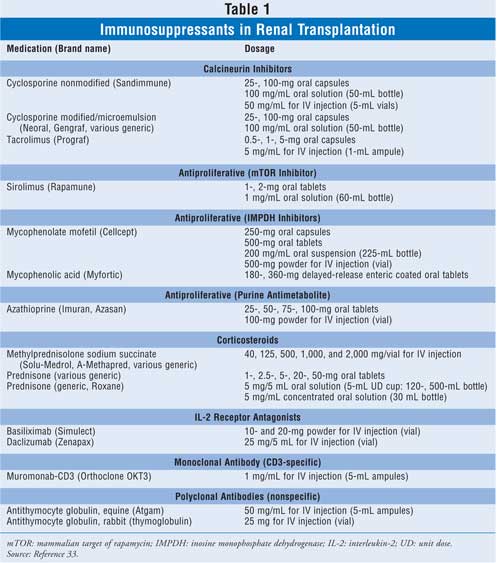
Immunosuppression in renal
transplantation (Table 1) typically consists of two phases: induction
and maintenance. Induction is utilized in the perioperative time period, when
the allograft (transplant, nonself) is at the greatest immunologic risk of
rejection. Induction immunosuppression is potent and involves using large,
tapering doses of corticosteroids (e.g., methylprednisolone), large doses of
calcineurin inhibitors (e.g., cyclosporine, tacrolimus), and in many cases, a
polyclonal (e.g., antithymocyte globulin) or monoclonal antibody (e.g.,
basiliximab, dacl izumab, or muromonab-CD3). Maintenance immunosuppression is
less intense than induction immunosuppression and is used for the life of the
allograft. Conventional maintenance immunosuppression involves a triple-drug
combination comprising a calcineurin inhibitor, an antiproliferative agent,
such as azathioprine, mycophenolic acid, or sirolimus, and in many cases, a
corticosteroid, such as prednisone. However, other combinations have been
utilized effectively.2
Corticosteroids exert their
immunosuppressive action by reducing cytokine synthesis in T and B lymphocytes
and through regulation of numerous cellular receptors that are important in
lymphocyte trafficking and communication.
Calcineurin inhibitors inhibit
the action of T lymphocytes by negating the action of calcineurin, a
phosphatase responsible for several intracellular events that lead to
increased synthesis of the proliferative cytokine interleukin-2 (IL-2).
The antiproliferative agents
differ from one another by their pharmacodynamics. Azathioprine is a purine
analog that acts by interfering with RNA and subsequent protein synthesis. The
agent is nonspecific and affects all rapidly dividing cells. Mycophenolic acid
is an inhibitor of the enzyme inosine monophospate dehydrogenase, resulting in
reduced purine synthesis in T and B lymphocytes. The drug is reported to be
more specific for lymphocytes because they rely on de novo synthesis of purine
bases, unlike other cells in the body that can utilize salvage pathways for
purine synthesis. Sirolimus is a target of rapamycin inhibitor (TOR). By
binding to TOR, sirolimus reduces the activity of the S6 ribosome and reduces
cellular protein synthesis, leading to cell cycle arrest from the G1-S phase.
IL-2 receptor antagonists are
bioengineered antibodies that limit the binding of IL-2 to the CD25 subunit of
the IL-2 receptor on T lymphocytes, reducing the intracellular events that
ultimately lead to proliferation of the T cell line.
Muromonab-CD3 (OKT3) is a
monoclonal antibody preparation that binds to the CD3 receptor on activated T
lymphocytes. OKT3 binding to the CD3 receptor causes a conformational change
and downregulation of the T cell receptor/CD3 complex, resulting in
circulating but inactive T cells.
Antithymocyte globulin
polyclonal antibody preparations consist of many different isoforms of
immunoglobulin G (IgG) antibodies that bind to numerous cellular receptors on
lymphocytes. Binding to various lymphocyte surface receptors results in
several pharmacodynamic effects, including inactivation or modulation of
receptors, complement-dependent or antibody-directed cellular lysis, and
receptor-activated programmed cell death or apoptosis.2
A recent article highlights
data compiled from the Scientific Registry of Transplant Recipients for the
time period of 1994 to 2003. For cadaveric renal transplant recipients with
nonexpanded donor criteria, one- and five-year graft survival rates were
reported as 91% and 69%, respectively. In the same time period, recipients of
living renal transplants had one- and five-year graft survival rates of 95%
and 79%. Factors that contributed to reduced allograft survival included
increased cold ischemia time (hours that the kidney is on ice during transport
or in storage prior to surgical placement), donor age over 65 years, delayed
graft function (need for dialysis during the first week of
posttransplantation), second transplant, African-American race, a higher
degree of human leukocyte antigen mismatch, and diabetes.1
Cardiovascular Disease (CVD)
CVD accounts for
the majority of deaths in renal transplantation. The incidence of CVD has been
reported to be as much as four times greater in renal transplant recipients
than that seen in the general population.4 Risk factors for CVD in
renal transplant patients are similar to, but more extensive than, the general
population. Patients with hypertension (HTN), diabetes, renal disease
(glomerular filtration rate <60 mL/minute or microalbuminemia), left
ventricular hypertrophy, and hyperlipidemia have a higher risk of CVD.
Patients who have a family history of premature CVD (men ages <55, women ages
<65) or history of acute cellular rejection also have an increased risk.
Obesity (body mass index [BMI] ?30 kg/m2), physical
inactivity, smoking, immunosuppression, and age (men ages >55, women ages >65)
are additional risk factors.3,4 The risk of CVD in kidney
transplant recipients can be attributed largely to preexisting CVD, length on
dialysis, type of immunosuppressive regimen, HTN, hyperlipidemia, and
posttransplant diabetes mellitus (PTDM).2-4
Hypertension
HTN is a
significant risk factor for CVD. It has been reported that the risk of CVD
doubles with each 20-mmHg increase in systolic blood pressure (SBP) or 10-mmHg
increase in diastolic blood pressure (DBP) between the ages of 40 and 70 years.
3 Mange et al.5 retrospectively studied the effect of HTN on
renal allograft survival in renal transplant patients. They found that the
rate of allograft failure increased by 15% for each 10-mmHg increase of SBP,
27% for every 10-mmHg increase in DBP, and 30% for every 10-mmHg increase in
mean arterial pressure.
Calcineurin inhibitors and
corticosteroids are the immunosuppressive agents most frequently implicated in
posttransplant HTN. There are several hypotheses regarding the mechanism of
these unwanted effects on blood pressure. Corticosteroids can induce the
retention of sodium in a dose-related fashion via their mineralocorticoid
activity and potency. Corticosteroids are also reported to elevate peripheral
vascular resistance by increasing vascular sensitivity to circulating
catecholamines.6 Cyclosporine (CSA) is believed to induce HTN by
enhancing afferent renal artery vasoconstriction, leading to activation of the
renin-angiotensin-aldosterone system, increasing sodium retention and
sympathetic nervous system discharge. Reduced vasodilatory prostaglandin
synthesis and enhanced vascular endothelin production are also reported
systemic mechanisms of CSA-induced HTN. Tacrolimus (FK506) shares many of the
same mechanisms for calcineurin-associated HTN but does not affect peripheral
resistance to the extent that CSA does, and HTN has been found to be less
severe in patients taking FK506, compared to CSA. Azathioprine, mycophenolate
mofetil, and sirolimus are generally considered to have a neutral effect on
blood pressure.2,7
In a two-week crossover study
in healthy volunteers, FK506 had little effect on renal hemodynamics and mean
arterial pressure, compared to CSA.8 In a follow-up of five-year
data from the phase III U.S. Multicenter Trial of tacrolimus versus CSA,
significantly more patients who received CSA required treatment with an
antihypertensive medication, compared to patients taking tacrolimus (91% vs.
81%; P < .05). All patients received a monoclonal (OKT3) or polyclonal
(Atgam) induction regimen and were maintained on azathioprine and prednisone
for the duration of the study. These results must be interpreted with caution,
since the hypertensive data may have been confounded by better graft function
in the patients randomized to FK506 versus CSA (median serum creatinine at
five years of 1.7 mg/dL versus 1.4 mg/dL; P =.0014).9
According to recent
recommendations for the prevention of posttransplant cardiovascular
complications, HTN should be treated as directed by the Seventh Report of the
Joint National Committee on Prevention, Detection, and Treatment of High Blood
Pressure (JNC VII) guidelines for HTN, since long-term outcome data is lacking
in renal transplant recipients.4 Transplant recipients should
initiate lifestyle changes that help control blood pressure and lower
cardiovascular risk (e.g., physical activity, smoking cessation, limiting
alcohol consumption, reducing salt intake). If lifestyle changes are not
effective, patients should be given drug therapy starting with stage I HTN
(SBP >140 mmHg or DBP >90 mmHg) or the prehypertension range (SBP >130 mmHg or
DBP > 80 mmHg) if patients have diabetes mellitus or chronic kidney disease.
Patients with stage II HTN (SBP >160 mmHg or DBP >100 mmHg) should be
initiated on two antihypertensive agents to control their blood pressure. In
each case, and unless there is a hypertensive emergency with target organ
damage, blood pressure should be reduced gradually to baseline blood pressure
and/or goal blood pressure. Compelling indications should be taken into
consideration when choosing antihypertensive therapy.3 Data
collected from the Collaborative Transplant Study database on long-term graft
and patient survival suggest that sustained blood pressure control (SBP <140
mmHg) can significantly increase patient and graft survival based largely on a
reduction in CVD.10
Beta blockers and
dihydropyridine calcium channel blockers (CCBs) are the most frequently
utilized agents in the immediate postrenal transplant phase. If no renal
artery stenosis or contraindications exist, angiotensin-converting enzyme
inhibitors (ACEIs) are chosen early on by some clinicians with careful
monitoring. Many clinicians add these agents to the regimen later on, when the
kidney transplant is functioning well and the serum creatinine has stabilized.
Clonidine, alpha blockers, and direct vasodilators are frequently used as
adjunct therapy or when contraindications exist for CCBs, ACEIs, or beta
blockers.2,3,11-13
There is some controversy
regarding the treatment of HTN in the early postoperative time period (first
24 to 48 hours). Recipients of cadaveric renal transplants may be prone to
allograft renal failure depending on cold ischemia time, immunologic risk, and
potential mechanical complications. Some clinicians suggest keeping patients
slightly hypertensive immediately after surgery to ensure that the allograft
is being adequately perfused.2 Others suggest that increased SBP in
the early postoperative phase may increase the risk of acute rejection due to
ischemia-reperfusion injury and have advised using antihypertensive agents to
keep SBP lower during this critical period.10 Thus, blood pressure
control in the immediate postoperative phase should be based on the available
data, in combination with clinician experience and extensive patient
monitoring.
Dyslipidemia
Hyperlipidemia is a
major risk factor for CVD in the renal transplant patient, as seen in a
post-hoc analysis of the Assessment of Lescol in Renal Transplantation (ALERT)
study, a large trial spanning five years that examined cardiovascular risk
factors and the effects of treatment with a hydroxymethyl glutaryl coenzyme A
(HMG-CoA) reductase inhibitor or a statin on cardiovascular events in renal
transplant patients. It was shown that total cholesterol (TC) and low-density
lipoprotein (LDL) were independent risk factors that significantly increased
the chances for nonfatal myocardial infarction (MI) (P <.01).14
In transplant patients randomized to 40 mg of fluvastatin and upward
titration to 80 mg (65% of treatment group), there was a 35% reduction in the
secondary endpoint of combined cardiovascular death and nonfatal MI, compared
to the placebo group (P =.005). Cardiovascular mortality alone did not
reach significance in ALERT. Since less than 10% of patients studied had a
history of CVD, ALERT was largely a primary prevention trial.15
Immunosuppressive agents and
regimens can be extremely effective in controlling the immune system and
preventing rejection, although they may promote unfavorable cholesterol and
triglyceride (TG) metabolism. The antiproliferative agents azathioprine and
mycophenolate mofetil have been reported to be lipid-neutral. The calcineurin
inhibitors can increase TC, LDL, and TG levels. CSA is much more potent at
inducing these changes than tacrolimus. Suggested mechanisms for
cyclosporine's effect include interfering with bile acid synthesis and
attaching to lipoproteins, altering the body's ability to regulate lipid
metabolism at the receptor level. Corticosteroids are also notorious for
increasing TC and TG levels-- an effect that may be additive when combined with
CSA or sirolimus. Corticosteroids are thought to increase the activity of
HMG-CoA and reduce the activity of lipoprotein lipase. Sirolimus can increase
TC levels and profoundly increase TG levels through various pathways
responsible for enhanced synthesis and reduced metabolism and clearance.16
However, it is not known if these increased values confer increased risk of
atherosclerotic CVD, since sirolimus has been successful in preventing vessel
proliferation when used in drug-eluting stents placed after angioplasty.
Emerging data suggests that systemic exposure to sirolimus does not increase
atherosclerotic CVD risk, even if TC and TG levels are high, and systemic
exposure to sirolimus may help prevent recurrent coronary in-stent restenosis.
17,18
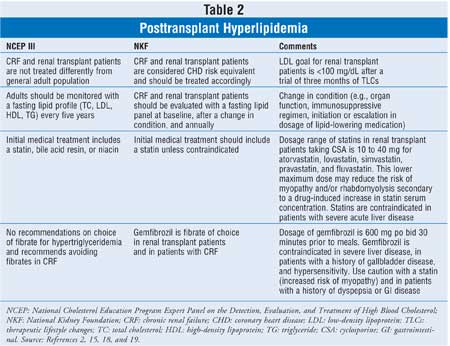
The National Kidney Foundation
(NKF) recently published clinical practice guidelines for managing
dyslipidemia in kidney transplant patients.19 These
guidelines expand on the National Cholesterol Education Program Expert Panel
on the Detection, Evaluation, and Treatment of High Blood Cholesterol (NCEP
III).20 Differences between the NKF guidelines and NCEP III are
summarized in Table 2. The NKF raises kidney transplant recipients to
the high-risk NCEP III category of coronary heart disease risk equivalent. The
lipid-monitoring (fasting TC, LDL, HDL, and TG) frequency is increased to
baseline, after every change in status (e.g., change in immunosuppression,
renal function, addition or change in lipid-lowering therapy,), and annually.
The following treatment recommendations assume that no contraindications,
prior adverse reactions, or drug interactions exist. They recommend statin use
in patients with LDL levels greater than 100 mg/dL after a trial of
therapeutic lifestyle changes (e.g., weight loss, low-fat diet, exercise) or
immediately in patients with LDL levels greater than 130 mg/dL. Patients with
isolated, high TG levels (TG > 500 mg/dL) should undergo therapeutic lifestyle
changes (TLC) and treatment with a fibrate (gemfibrozil) to prevent
pancreatitis, in addition to atherosclerotic CVD. Non-HDL cholesterol (TC-HDL)
should be used as a target for lipid lowering in patients with TG levels
higher than 200 mg/dL, even if their LDL level is low or normal, because
higher levels have been associated with the metabolic syndrome and
atherosclerotic CVD. The treatment targets for non-HDL are essentially 30
mg/dL higher than the LDL target ranges set forth in the NCEP III. Thus, CHD
risk equivalent patients (kidney transplant patients) should receive drug
treatment at non-HDL values higher than 160 mg/dL and at non-HDL values
greater than 130 mg/dL after a trial of therapeutic lifestyle changes.
If a statin is prescribed,
clinicians must utilize proper monitoring for efficacy and safety with a lipid
panel at baseline and every four to six weeks, in addition to liver function
tests at baseline, 12 weeks, and semiannually. The most common drug
interaction between an immunosuppressive agent and a statin involves CSA. When
given concurrently with CSA, statins marginally affect the area under the
curve of CSA. CSA can significantly increase the area under the curve of some
statins, increasing the risk of myopathy and rhabdomyolysis. Mechanisms
thought to be responsible for this interaction include inhibition of
cytochrome P-450 3A4 (CYP3A4), competition with biliary elimination, and
inhibition of gut transport via p-glycoprotein or the
multidrug-resistance-associated protein-2. Due to the correlation of these
interactions with statin dosage, it is generally recommended that the maximum
dosage of statins be lowered by 50% if used in conjunction with CSA (i.e., 40
mg for atorvastatin, lovastatin, pravastatin, simvastatin, and fluvastatin).
Due to ALERT, fluvastatin is the best-studied statin in the renal transplant
population with a benign side-effect profile, even when the dose was bumped to
80 mg in a large number of patients. Patients may be at a greater risk for
liver disease, myopathy, and rhabdomyolysis when statins are prescribed in
conjunction with fibrates or niacin and will require intense monitoring. It is
extremely important to remind patients to report any muscle pain/weakness to
their transplant physician, especially if the statin is a new medication, if
an increase in dosage has been prescribed, if CSA is part of the
immunosuppressive regimen, or if a fibrate or niacin is being added to
concurrent statin therapy.14,16,19,20
If a patient's TG value does
not respond optimally to statin therapy titrated to an acceptable dose for
kidney transplant recipients, a fibrate (e.g., gemfibrozil) may be added,
provided there are no contraindications, such as liver disease, and there is
close monitoring for adverse effects (e.g., liver function test abnormalities,
myopathy, gastrointestinal problems). However, the statin/fibrate combination
has not been shown to dramatically reduce TG and TC levels, compared to a
statin alone. Fibrates can also be utilized for the treatment of isolated,
very high TG levels (>500 mg/dL), which is often seen with patients taking
sirolimus. Gemfibrozil is the recommended fibrate to use in the renal
transplant population because the dose does not need to be adjusted in
patients with renal insufficiency/failure. Niacin may be used as a second-line
agent in patients who have failed to achieve their treatment goal with optimal
dosing of statins and/or fibrate therapy. Niacin has a beneficial effect on
all lipid parameters but is associated with many adverse effects. These
include hyperglycemia, hyperuricemia, flushing, liver toxicity,
gastrointestinal intolerance, and increased risk of myopathy when combined
with a statin. Niacin has not been studied extensively in renal transplant
patients. Contraindications to niacin include liver disease, gout, and upper
gastrointestinal bleeding/ulceration. Bile acid sequestrants like
cholestyramine have not been utilized often in renal transplant patients due
to gastrointestinal adverse effects, their effect on TG levels, and the
potential for binding immunosuppressive and other critical medications in the
gut, limiting their absorption.16,19,20
Posttransplant Diabetes
Mellitus
Diabetes is
frequently diagnosed early after renal transplantation and contributes
significantly to CVD. In addition, renal transplant patients with PTDM have
lower graft and patient survival rates and less renal function over time, as
demonstrated by higher serum creatinine values, compared to renal transplant
patients without PTDM.21,22 In a published review of 11,659 first
kidney renal transplant recipients with Medicare as their primary form of
insurance from 1996 to 2000, PTDM was diagnosed in 9.1%, 16%, and 24% of
patients within three, 12, and 36 months of transplantation. Most of the
patients studied received a corticosteroid, a calcineurin inhibitor (CSA or
FK506), and mycophenolate mofetil.23 Recently, the incidence
of PTDM has been reported to be as high as 74% within the year following
transplantation.24
Factors that have been shown
to significantly increase the likelihood for PTDM include, but are not limited
to, immunosuppressive medications (corticosteroids and calcineurin
inhibitors), age above 40 years, race (African-American, Hispanic), body
weight (BMI > 30 kg/m2), male donor, degree of human leukocyte
antigen mismatch, hepatitis C infection, and family history of diabetes.23
Clinicians can help modify or reduce these risk factors by educating patients
about weight loss and the effects of a well-rounded diet and benefits of
exercise and by using immunosuppressive strategies that lessen the risk of
PTDM and impaired glucose tolerance.25
The type of immunosuppression
can have a dramatic effect on the incidence of PTDM. Clinicians continue to
study methods to reduce the risk of PTDM without increasing the chances for
allograft rejection. Corticosteroids have been shown to contribute to glucose
intolerance in a dose-dependent and time-dependent manner by reducing
peripheral insulin sensitivity and marginally reducing insulin production.
There is a large amount of published literature on successfully reducing PTDM
by using corticosteroid avoidance, withdrawal, and dosage reduction
strategies. By using one of these methods, the risk for allograft rejection
and consequences must always be taken into consideration by the transplant
physician prior to implementation, and the patient must be intensely monitored.
25,26
The effects of calcineurin
inhibitors (CSA and tacrolimus) on PTDM are well described in the literature.
A study was recently published as a follow-up report on five-year data from
the phase III U.S. Multicenter Trial of Tacrolimus in Kidney Transplantation.
Four hundred and twelve patients were randomized to CSA or FK506 dosed to
standard trough concentrations at three months (100 to 300 ng/mL for CSA, 5 to
15 ng/mL for FK506), a maintenance regimen consisting of prednisone tapered to
10 mg/day by six months and azathioprine. All patients received OKT3 or Atgam
for induction immunosuppression. After one year, 19.9% of patients taking
FK506 had PTDM, compared to 4% of patients taking CSA. There was a large
degree of reversal in the FK506 arm; 41.2% of patients taking FK506 were able
to discontinue insulin after five years, compared to 14% of patients
randomized to CSA.9 A recent meta-analysis involving PTDM in
transplantation compared CSA to FK506, and the incidence was somewhat lower
than reported above (9.8% for FK506, 2.7% for CSA; P <.00001). The authors
classified patients as having PTDM when insulin was administered, which may
have accounted for the lower numbers.27 CSA is hypothesized to
contribute to PTDM by interfering with corticosteroid metabolism, leading to
reduced peripheral insulin sensitivity. CSA also inhibits insulin synthesis
and secretion from pancreatic beta cells. Tacrolimus shares the same reported
mechanism but may be more potent in its ability to inhibit insulin synthesis
and secretion. This may be related to differences in pancreatic intracellular
CSA and FK506 binding proteins (cyclophylin and FKBP-12). The effects of both
calcineurin inhibitors on insulin production and secretion may be dose
related, as CSA has been shown to be more diabetogenic in animals with higher
CSA blood levels, and FK506-induced PTDM has been correlated with higher
trough concentrations in human renal transplant patients.25,26,28
There have been many studies published recently on calcineurin avoidance,
withdrawal, and dosage sparing that have reported successful reduction in PTDM
incidence rates without increasing the risk of rejection in renal
transplantation. Many of these studies employed antibody induction with an
IL-2 antagonist or a polyclonal antibody preparation (Thymoglobulin).
Sirolimus has been used in many of these studies with some success. The
optimal approach remains to be determined.
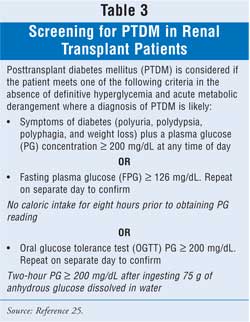
The diagnosis of PTDM has been
refined and standardized by the 2003 International Consensus Guidelines for
New-Onset Diabetes after Transplantation (Table 3). This document is
based collectively on U.S. and international guidelines for the diagnosis and
management of diabetes in the general population; recommendations included the
best published literature on PTDM. Unfortunately, PTDM was defined differently
in many of the studies published. Some studies defined PTDM by the duration of
insulin use, others used impaired fasting glucose and/or oral glucose
tolerance testing values, and a few included patient use of oral hypoglycemic
medication posttransplantation. This makes it very difficult to examine the
impact that various immunosuppressive medications have on the incidence of
PTDM across studies. Fasting plasma glucose should be obtained every week
immediately following transplantation, three, six, and 12 months, and annually
thereafter. Plasma glucose should also be monitored frequently during the
immediate posttransplant period, when patients are receiving high doses of
corticosteroids and calcineurin inhibitors.21,25
Once PTDM is diagnosed,
patients should self-monitor their glucose at home if possible to examine how
their plasma glucose level responds to carbohydrate intake, lifestyle
alterations (e.g., diet, exercise), and drug therapy. It is recommended that
HgbA1c be obtained every three months with a target value
less than 6.5% (normal 4% to 6%) based on data suggesting that tight glycemic
control lessens the progression of microvascular and macrovascular
complications. Patients should be taught how to recognize complications that
may develop secondary to their disease progression (e.g., neuropathy,
retinopathy, nephropathy, infection) and should be monitored by their
physician annually.25
Prior to initiating drug
therapy and depending on the severity of PTDM, transplant physicians may
change the immunosuppressive regimen by reducing the corticosteroid dose or
withdrawing the steroid altogether if the immunological risk is low.
Consideration may also be given to reducing the dose of the calcineurin
inhibitor or switching from tacrolimus to CSA or from a calcineurin inhibitor
to sirolimus. With tacrolimus, early PTDM is likely to be transient or may be
less severe when the dosage is reduced to maintain lower trough blood levels.
All of the above must be performed under close supervision of the transplant
physician due to the potential for acute rejection. A dietician should be
consulted in patients at risk for PTDM to help with optimal dietary lifestyle
changes and to help with calorie restriction and weight reduction in patients
that require such changes. Patients should be educated on the benefits and
importance of exercise, even if this consists of walking for 30 minutes
per day. Education regarding optimal smoking cessation strategies should also
be provided.25-30
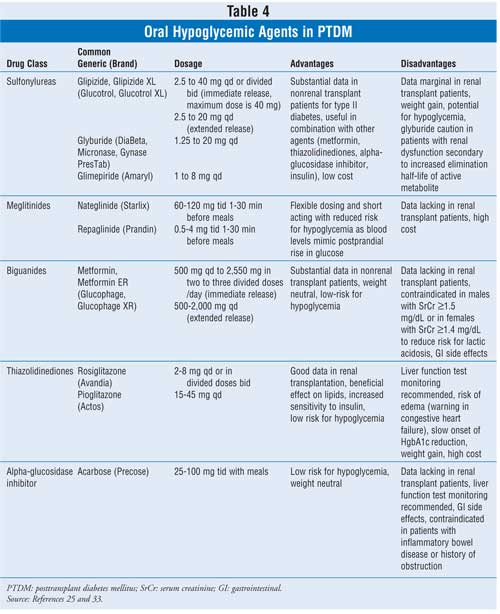
Surprisingly, there are few
large prospective studies on the use of oral hypoglycemic agents and insulin
in renal transplant patients. The consensus guidelines recommend an oral
hypoglycemic medication as the initial agent of choice, followed by an oral
hypoglycemic combination, an oral hypoglycemic agent with insulin, or with
insulin alone.25 Patients with very high levels of plasma glucose
may require immediate treatment with insulin, especially in the early
postoperative period or in situations when patients are acutely ill (e.g.,
infection, other surgery). When an oral hypoglycemic agent is utilized in
renal transplant patients, care must be taken regarding differences between
the agents, contraindications, and the available evidence for use in this
population based on safety and efficacy (Table 4).
Two recent prospective studies
31,32 were published examining the use of the thiazolidinediones
(rosiglitazone and pioglitazone) in the management of PTDM in renal and liver
transplant patients. The thiazolidinediones are peroxisome
proliferator-activated receptor gamma agonists that reduce insulin resistance
in skeletal muscle. Both agents were effective in reducing insulin
requirements and lowering HgbA1c to less than 6.5%, often in combination with
a sulfonylurea. Patients with congestive heart failure were excluded, and no
drug interactions were noted. The most frequent adverse effect reported in the
two trials was edema. Despite thiazolidinediones' long onset of action and
potential to cause edema, the authors cited the beneficial effects that they
have on levels of LDL, HDL, TG, and the vascular endothelium that may make
these agents advantageous in the management of PTDM.25
Conclusion
Cardiovascular complications in renal transplant patients can be reduced or prevented with optimal pharmacological management and follow-up care. This is a relatively new and exciting area of pharmacotherapy. A substantial amount of data that will impact the way we care for renal transplant patients is continuing to evolve and is becoming even more complex. Pharmacists have an important role in kidney transplantion, as they foster patient compliance through education, are frequently accessible, have the ability to look for prescription and over-the-counter drug interactions, monitor for adverse reactions, serve as a resource for the transplant team physicians and nurses in hospital and ambulatory environments, and have a wide variety of knowledge at their fingertips.
References
1. Danovitch GM, Cohen DJ, et al. Current status of kidney and pancreas transplantation in the United States, 1994-2003. Am J Transplant. 2005;5:904-915.
2. Danovitch GM. Handbook of Kidney Transplantation. 3rd ed. Philadelphia: Lippincott Williams & Wilkins; 2001:163-220.
3. Chobanan AV, Bakris GL, et al. The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High blood pressure (The JNC 7 Report). JAMA . 2003;289:2560-2572.
4. Bostom AD, Brown RS, et al. Prevention of posttransplant cardiovascular disease – report and recommendations of an ad hoc group. Am J Transplant. 2002;2:491-500.
5. Mange KC, Cizman B, et al. Arterial hypertension and renal allograft survival. JAMA. 2000;283:633-638.
6. Brem AS. In-depth review: insights into glucocorticoid-associated hypertension. Am J Kidney Dis. 2001;37:1-10.
7. Miller LW. Cardiovascular toxicities of immunosuppressive agents. Am J Transplant. 2002;2:807-818.
8. Klein IH, Abrahams A, et al. Different effects of tacrolimus and cyclosporine on renal hemodynamics and blood pressure in healthy subjects. Transplantation. 2002;73:732-736.
9. Vincenti F, Jensik SC, et al. A long-term comparison of tacrolimus (FK506) and cyclosporine in kidney transplantation: evidence for improved allograft survival at five years. Transplantation 2002; 73:775-782.
10. Opelz G, Dohler B. Improved long-term outcomes after renal transplantation associated with blood pressure control. Am J Transplant. 2005;5:2725-2731.
11. Thomas MC, Mathew TH, et al. Perioperative blood pressure control, delayed graft function, and acute rejection after renal transplantation. Transplantation. 2003;75:1989-1995.
12. Taler SJ, Textor SC, et al. Cyclosporin-induced hypertension. Drug Safety. 1999;20:437-449.
13. Olyaei AJ, DeMattos AM, et al. A practical guide to the management of hypertension in renal transplant recipients. Drugs. 1999;58:1011-1027.
14. Jardine AG, Fellstrom B, et al. Cardiovascular risk and renal transplantation: post hoc analysis of the Assessment of Lescol in Renal Transplantation (ALERT) study. Am J Kidney Dis . 2005;46:529-536.
15. Holdaas H, Fellstrom B, et al. Effect of fluvastatin on cardiac outcomes in renal transplant recipients: a multicenter, randomized, placebo-controlled trial. Lancet. 2003;361:2024-2031.
16. Mathis AS, Dave N, et al. Drug-related dyslipidemia after renal transplantation. Am J Health-Syst Pharm. 2004; 61:565-587.
17. Cheuh SJ, Kahan BD. Dyslipidemia in renal transplant recipients treated with a sirolimus and cyclosporine-based immunosuppressive regimen: incidence, risk factors, progression, and prognosis. Transplantation. 2003;76:375-382.
18. Hausleiter J, Kastrati A, et al. Randomized, double-blind, placebo-controlled trial of oral sirolimus for restenosis prevention in patients with in-stent restenosis: the oral sirolimus to inhibit recurrent in-stent stenosis (OSIRIS) trial. Circulation. 2004;110:790-795.
19. Kasiske B, Cosio FG, et al. Clinical practice guidelines for managing dyslipidemias in kidney transplant patients: a report from the managing dyslipidemias in chronic kidney disease work group of the National Kidney Foundation kidney disease outcomes quality initiative. Am J Transplant. 2004;4(Suppl. 7):13-53.
20. Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive Summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA. 2001;285:2486-2497.
21. Montori VM, Basu A, et al. Posttransplantation diabetes: a systematic review of the literature. Diabetes Care. 2002;25:583-592.
22. Cosio FG, Pesavento TE, et al. Patient survival after renal transplantation: IV. Impact of posttransplant diabetes. Kidney Int. 2002;62:1440-1446.
23. Kasiske BL, Snyder JJ, et al. Diabetes mellitus after kidney transplantation in the United States. Am J Transplant. 2003;3:178-185.
24. Sulanc E, Lane JT, et al. New-onset diabetes after kidney transplantation: an application of 2003 international guidelines. Transplantation. 2005;80:945-952.
25. Davidson J, Wilkinson A, et al. New-onset diabetes after transplantation: 2003 international consensus guidelines. Transplantation. 2003;75(10 Suppl):SS3-SS24.
26. Weir MR, Fink JC. Risk for posttransplant diabetes mellitus with current immunosuppressive medications. Am J Kidney Dis.1999;34:1-13.
27. Heisel O, Heisel R, et al. New onset diabetes mellitus in patients receiving calcineurin inhibitors: a systemic review and meta-analysis. Am J Transplant. 2004;4:583-595.
28. Nam JH, Mun JI, et al. ‚ Beta-cell dysfunction rather than insulin resistance is the main contributing factor for the development of postrenal transplantation diabetes mellitus. Transplantation. 2001;71:1417-1423.
29. Mulay AV, Hussain N, et al. Calcineurin inhibitor withdrawal from sirolimus-based therapy in kidney transplantation: a systematic review of randomized trials. Am J Transplant . 2005;5:1748-1756.
30. VanHooff JP, Christiaans MH, et al. Tacrolimus and posttransplant diabetes mellitus in renal transplantation. Transplantation. 2005;79:1465-1469.
31. Villanueva G, Baldwin D. Rosiglitazone therapy of posttransplant diabetes mellitus. Transplantation . 2005;80:1402-1405.
32. Luther P, Baldwin D. Pioglitazone in the management of diabetes mellitus after transplantation. Am J Transplant. 2004;4:2135-2138.
33. Facts and Comparisons Electronic
(Version 4.0). Wolters Kluwer Health, Inc. 2005.
To comment on this article, contact
editor@uspharmacist.com.





