US
Pharm. 2006;6:62-70.
According to the United States Census
Bureau, in July 2003, there were approximately 62 million women of
childbearing age in the U.S.1 Forty-three million (69%) of these women were
fertile, sexually active, and did not desire to become pregnant.2 These
statistics help portray the high public demand for contraceptive agents.
Contraceptive products comprise oral,
transdermal, intravaginal, intrauterine, intramuscular, and subcutaneous
preparations available in a variety of doses and estrogen-progestin
combinations. Due to the extent of products offered, it can be difficult for
health care providers to stay up-to-date on and understand the advantages and
disadvantages of different dosage forms. This article provides an overview of
hormonal contraceptive products and briefly discusses the risks and benefits
associated with specific dosage forms to assist providers in the selection of
the most appropriate product.
Estrogens
There are two types of estrogen
found in contraceptives. The more common type is ethinyl estradiol. A few oral
contraceptive products contain mestranol, which is metabolized by the liver to
ethinyl estradiol. Mestranol is estimated to have half the potency of ethinyl
estradiol.3 Estrogens are an important component of contraceptive
products and work by preventing the formation of a follicle. When given in
amounts that exceed normal endogenous levels, a negative feedback loop is
created between the hypothalamus and anterior pituitary. This negative
feedback loop prevents the release of follicle-stimulating hormone (FSH) from
the anterior pituitary. When FSH levels are kept low, a follicle is not able
to form and ovulation is prevented.3-5
Progestins
Progestins are classified into
first, second, third, and fourth (or spironolactone-derived)
generations. Progestin generations can be quite confusing, as some studies
classify them based on the date that the progestin was introduced to the
market, while others classify them according to the progestin's carbon-ring
structure. Therefore, the same progestin can be classified in different
generations depending on the reference used.6 For this reason, as
well as the wide variation of side effects seen with progestins within the
same generation, this article discusses progestins according to their degree
of estrogenic, androgenic, and progestational activity.
Progestins are effective contraceptive
agents when used alone or in combination with an estrogen product. When used
in combination with an estrogen, progestins inhibit the luteinizing hormone
(LH) surge, which is required for ovulation. When progestins are used alone,
they are not thought to consistently inhibit ovulation, possibly because of
the significantly lower doses of progestin in progestin-only contraceptives as
compared to combination products.4 Progestin-only contraceptives
are thought to function primarily by creating a hostile cervical environment
(thickening of the mucus) and causing endometrial atrophy. These changes
inhibit egg implantation and decrease the penetration of sperm and ovum
transport.3-8
Each progestin is associated with a slightly
different affinity for estrogen, androgen, and progesterone receptors (table 1)
, resulting in the various side effects seen with each progestin. Side effects
for different progestins are discussed later in this article.

Preparations
Formulations of hormonal
contraceptives vary from traditional oral tablets and once-weekly patches to
intrauterine devices that are effective for up to five years after
implantation. When selecting a contraceptive product, patient preference,
patient tolerance, the various adverse effect profiles, and the formulations
available should all be considered in order to maximize efficacy and patient
adherence.
Oral Contraceptives:
The efficacy of oral contraceptives with perfect use is above 99%.3,9
This can be misleading to patients because perfect use implies that no doses
are missed, the pill is taken at the same time every day, and appropriate
backup methods are used when indicated. Studies have found that the efficacy
rate drops to approximately 95% for typical use, which accounts for patient
noncompliance and inappropriate use (e.g., no backup method used when
indicated).3,6,8-10
Of all hormonal contraceptive dosage forms,
oral preparations are available in the largest variety of products. Oral
formulations differ with regard to the dose, as well as combination, of
progestin and estrogen they contain. Newer formulations of oral contraceptives
contain approximately one fifth the amount of estrogen and one tenth the
amount of progestin compared to older preparations.3,6,8 Currently,
oral combination contraceptive products contain ethinyl estradiol in doses
ranging from high-dose preparations with 50 mcg to ultra-low-dose formulations
containing 20 mcg. Average ethinyl estradiol doses range from 30 to 35 mcg.
Products that consist of mestranol have 50 mcg of the hormone, which has been
found to be equivalent to approximately 35 mcg of ethinyl estradiol.11,12
Estrogen doses were initially lowered after reports of increased risk of
ischemic stroke, myocardial infarction, and pulmonary embolism with high-dose
estrogen preparations.9,12-14 Doses have continued to decrease
because contraceptive efficacy has not diminished with lower-dose formulations.
7,9
Alterations have also been made to initial
oral combination products in order to more closely mimic a woman's natural
menstrual cycle. These formulations vary the dose of estrogen, progestin, or
both throughout the cycle and are called biphasic and triphasic preparations (
table 2).3,9,15 Monophasic preparations (table 3) are still
available and contain a consistent amount of estrogen and progestin in each
active tablet.

Yasmin (ethinyl estradiol/drospirenone) is a
combination, monophasic oral contraceptive that is unique in that its
progestin is structurally similar to spironolactone.7,15 Each
tablet contains 30 mcg of ethinyl estradiol and 3 mg of drospirenone, which is
equivalent to 25 mg of spironolactone.15 Due to the weak
potassium-sparing diuretic effect that drospirenone exhibits, this product may
be preferred by women who experience water retention associated with their
menstrual cycle. Patients should be monitored for hyperkalemia while on Yasmin
because of the anti mineralocorticoid properties of drospirenone.
7,15

An alternative to combination formulations is the
progestin-only pill (POP), or mini pill. Progestin-only pills provide an oral
alternative for lactating women, as progestins, unlike combination products,
have not been shown to decrease milk production.4,5,8 POPs are also
good options for women with contraindications or intolerance to
estrogen-containing contraceptives and for women who would like to become
pregnant in the near future, as progestin-only pills do not stop ovulation.
Strict adherence to timing of daily dosing is necessary with progestin-only
pills due to the decreased effect of the progestin on the cervical mucosa 22
hours after the dose is taken. A dose that is three hours late is considered
missed and should be taken as soon as it is remembered. Literature suggests
using alternative means of contraception for two days following late or missed
doses.3,5,6,8,9 POPs should be taken continuously with no pill-free
interval.
Progestin-only pills are slightly less
effective than combination oral contraceptives. With perfect use, POPs have an
efficacy rate of 99.5% compared with 99.9% for combination products.3,9
POPs are also associated with a greater degree of irregular bleeding.
Approximately 35% to 45% of women on POPs experience spotting and breakthrough
bleeding throughout the first three months of use; in comparison, 5% to 20% of
women using combination oral contraceptives experience these adverse effects.
3,9 Other common side effects of POPs include headache, breast
tenderness, nausea, and dizziness.9
Transdermal Contraceptives:
The delivery of hormones through transdermal means was traditionally used for
the administration of estrogen and testosterone. It was not until 2002 that
Ortho Evra (ethinyl estradiol/norgestimate), a combination estrogen and
progestin transdermal patch, became available as a form of contraception in
the U.S. The patch's mechanism of action is similar to that of oral
contraceptive pills in that 20 mcg of ethinyl estradiol and 150 mcg of
norelgestromin, a metabolite of norgestimate, are released daily.15,16
When compared with perfect use of daily oral
contraceptives, the patch had similar contraceptive efficacy (approximately
99%) for most patient populations. Transdermal contraceptives have an
advantage over oral preparations in that compliance is enhanced because of
once-weekly administration.17 In 2005, FDA added a bolded warning
to Ortho Evra's prescribing information stating that the pharmacokinetic
profile of Ortho Evra differs from that of the oral contraceptive pill.16
Specifically, compared to the pill, Ortho Evra exposes patients to higher
steady state concentrations and lower peak concentrations of ethinyl
estradiol. While increased estrogen exposure may increase the risk of serious
adverse events, it is not known whether Ortho Evra is associated with a higher
risk of adverse events compared to the oral contraceptive pill. Furthermore,
Ortho Evra has been associated with higher rates of unintended pregnancies in
patients weighing over 198 pounds (98 kg) and should therefore be used with
caution in this population.6,15,16 Studies have reported increases
in incidence of breast tenderness, poor cycle control, and application-site
reactions with Ortho Evra when compared with oral preparations.15,16
Ovulation rapidly returns for a majority of patients after discontinuation of
Ortho Evra, with most able to become pregnant within three months. For women
with irregular periods prior to therapy, pregnancy may be delayed.15
The Ortho Evra patch is applied to the
buttock, abdomen, upper outer arm, or upper torso on the first day of the
patient's menstrual period. A new patch is applied every week for three weeks,
followed by one patch-free week. Location of the patch should be rotated each
week to help reduce the incidence of skin irritation. If the patch becomes
partially or completely detached for less than one day, a new patch should be
applied or the same patch can be used if it still contains its adhesive, and
no backup method would be necessary. If the patch has been off for longer than
a day, a new patch should be placed immediately and the new "patch change day"
should be based on the day the new patch was applied. When the patch has been
off for more than 24 hours, a backup means of contraception is recommended
until the new patch has been on for seven consecutive days. A patch should not
be reapplied if it has stuck to itself or to another surface.15,16
Intrauterine Device:
There are two types of intrauterine devices (IUDs) approved in the U.S. The
more commonly used IUD is a copper-containing system. Copper-containing IUDs
are hypothesized to function by decreasing sperm's ability to reach the
uterine cavity and fallopian tubes for fertilization, as well as by creating
cellular changes to sperm that cause it to be nonviable.3-5
Efficacy rates for copper IUDs are over 99%. IUDs are associated with
increased incidence of endometritis, pelvic infections, allergic skin
reactions, tubal damage, and uterine perforation. One advantage of
copper-containing IUDs is that the side effects associated with estrogen and
progestins are avoided because it is a nonhormonal contraceptive product.
Other IUDs release hormones; these are
Progestasert (progesterone) and Mirena (levo norgestrel). Progestasert was
the first hormonal IUD, and it functions as a contraceptive by releasing
progesterone and therefore altering the cervical environment. The Progestasert
system is inserted by a physician and left in place for 12 months. The system
must be replaced every 12 months to ensure contraceptive effectiveness.
5,18 Progestasert is associated with a higher risk of unexpected
pregnancy when compared with other IUD systems and has an efficacy rate of
98%. Because of decreased efficacy and the need for yearly replacement,
Progestasert is not widely used.19 Common adverse effects of
Progestasert include intermenstrual spotting, and severe but rare effects
include pelvic inflammatory disease (PID), fragmentation of device, and
cervical perforation.4
The newest IUD available in the U.S., Mirena
exerts its contraceptive effects by releasing 20 mcg of levonorgestrel daily.
5,15,20 Ovulation has been shown to be prevented in some cycles, but
this system primarily functions secondary to progestin activity in the cervix.
19 Mirena has been proven over 99% effective for the five years
following insertion. This system is associated with increased risks of PID and
irregular bleeding but has been found to be associated with fewer
complications when compared to copper-containing devices.5,15,19,20
Return to ovulation is immediate upon
removal of IUDs, and approximately 80% of women are able to become pregnant
within one year.4,19 IUDs may be appealing options for women who
desire contraception over a long period of time.
Vaginal Contraceptive Ring:
The vaginal contraceptive ring, NuvaRing (ethinyl estradiol/etonogestrel),
works by delivering ethinyl estradiol at a rate of 15 mcg per day and
etonogestrel, the active metabolite of deso norgestrel, at a rate of 120 mcg
per day.15,20,21 Studies have shown that the Nuva Ring is 98% to
99% effective in preventing pregnancy and is generally well tolerated.
11,15,19,21 The NuvaRing is associated with a higher incidence of
vaginitis, vaginal discomfort, and vaginal infections when compared to other
forms of hormonal contraception and is not recommended for patients with
cystocele, rectocele, or uterine prolapse.15,21
NuvaRing is a flexible ring that is inserted into
the vagina by the fifth day of the menstrual cycle and is left in place for
three weeks. After three weeks, the ring is removed for one week and a new
ring is inserted at approximately the same time of day that the old ring was
removed.15,21 If the NuvaRing is out of the vagina for less than
three hours at any time during active therapy, the ring should be rinsed and
reinserted and no backup method is necessary. If the ring is out of the vagina
for more than three hours, a backup method should be initiated for the next
seven days, as contraceptive efficacy may be diminished. Return to ovulation
with the Nuva Ring is immediate, with ovulation commonly occurring within one
month following discontinuation of the product.21
Intramuscular (IM) Injections:
Depo-Provera (medroxyprogesterone acetate) was the first non oral hormonal
contraceptive agent available in the U.S.11 Medroxyprogesterone 150
mg is injected into the deltoid or gluteal muscle every three months and
functions by suppressing ovulation and altering the cervical environment.
5,22 The first injection must be given within the first five days of
menstruation to ensure the patient is not pregnant. Efficacy rates are
comparable to those with perfect use of oral contraceptive agents, and the
drug is effective 24 hours after the injection is given, provided it is given
within the first week of menses.21 Because injections are given
once every three months, compliance issues seen with other contraceptive
formulations are avoided, therefore yielding greater efficacy when comparing
typical use of hormonal contraceptive products.22
Common adverse effects of Depo-Provera
include irregular bleeding, weight gain, delays in return of ovulation, and
possible reductions in bone mineral density, especially in smokers.22
Studies have shown that ovulation most often recurs six to 12 months after
product discontinuation, with a median time of approximately seven months.
Duration of use does not seem to be related to length of time to recurrence of
ovulation.5,22
Subcutaneous Injections: In
December 2004, Depo-SubQ Provera 104 received FDA approval for use as a
contraceptive agent. By slightly altering the formulation of Depo-Provera, the
first subcutaneous hormonal contraceptive product was created. Depo-SubQ
Provera 104 is a suspension that contains 104 mg of medroxy progesterone
acetate and comes packaged in a prefilled syringe. A subcutaneous injection is
administered into the anterior thigh or the abdomen every three months and
functions as a means of contraception by preventing ovulation and causing
thinning of the endometrium. Results of three studies, which included more
than 2,000 women collectively, reported no unintended pregnancies throughout
the one-year follow-up period.23
Common adverse effects of Depo-SubQ Provera
104 are similar to those of Depo-Provera and include weight gain, injection
site reactions, irregular bleeding, loss of bone mineral density, and delays
of return to fertility. The median time to return of ovulation after product
discontinuation is approximately 10 months.23 One advantage of the
subcutaneous formulation when compared with the IM injection is ease of
administration, and home administration may be appropriate and would save the
patient from quarterly physician visits.
Risks of Hormonal Contraceptive Use
Common Adverse Effects:
Adverse effects are a common reason for discontinuation of hormonal
contraceptive agents and often produce confusion for health care providers who
need to determine the cause and find an appropriate therapeutic alternative.
One reason side effects vary between products is the diversity of estrogen and
progestin doses used. For example, women experiencing nausea, vomiting,
bloating, decreased libido, and breast tenderness may be receiving too much
estrogen and may benefit from a contraceptive with a lower estrogen dose.
3,8,18 Conversely, acne, hirsutism, and vaginal yeast infections have
been associated with excess amounts of progestin, and these effects can be
minimized by choosing an alternative product with a lower progestin content.
3,8,18 Common symptoms of estrogen and progesterone overload and
deficiencies are found in table 4.

Side effects can also be associated with specific
progestins, as each progestin has slight variations in its estrogenic,
androgenic, or progestational activity (table 1). For example, levonorgestrel
and norgestrel are progestins with high androgenic activity and are more prone
to cause acne, hirsutism, weight gain, fatigue, and depression than progestins
with less androgenic activity. By switching to a contraceptive with a less
androgenic progestin, these side effects may subside. The side effects
associated with high levels of estrogenic, progestational, and androgenic
activity are found in Table 5.

Serious Adverse Effects:
Despite the widespread use of hormonal contraceptive agents, there are some
serious risks involved. Combination contraceptives have been associated with
increased incidence of breast cancer, cervical cancer, myocardial infarction,
stroke, and venous thromboembolism, including deep venous thrombosis (DVT) and
pulmonary embolism.6,12,14,15 However, the overall risk for
experiencing adverse events with hormonal contraceptive products is small and
is closely associated with the woman's health status and presence of
cardiovascular risk factors, as well as the agent's estrogen dose. Patients
who smoke, are obese, have a family history of coronary artery disease, or are
over 35 years of age, as well as women with concomitant disease states,
including hypertension, diabetes, and hyperlipidemia, have been shown to be at
significantly greater risk of adverse events when compared with healthy women.
6,12,14,15 Thus, a thorough patient history should be obtained when
initially selecting a hormonal contraceptive agent, in order to evaluate the
presence of contraindications (table 6) and risk factors.
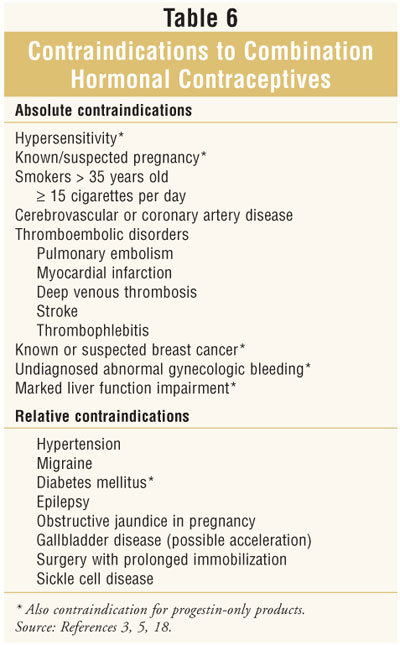
A majority of the risks involved with hormonal
contraceptives have been associated with combination products. As shown in
table 6, many of the typical risk factors that are contraindications for
combination products do not apply to progestin-only products. This is largely
because progestins are not associated with the cardiovascular complications
seen with estrogen use. Therefore, progestin-only products are good
alternatives for women at high risk for adverse events or those with
contraindications to estrogen.5,8,15
All patients started on hormonal
contraceptives should be counseled on the signs and symptoms of ACHES: A
bdominal pain, Chest pain, Headache, Eye pain, and Severe leg pain.3,8
The presence of these symptoms may be warning signs of stroke, hypertension,
pulmonary embolism, DVT, or gallbladder disease and require further assessment
to identify the cause.3,8
Noncontraceptive Benefits of Oral
Contraceptive Use
In spite of perceived risks of
contraceptive products, there are many benefits associated with their use.
Oral contraceptives have been shown to decrease the risk of ectopic pregnancy,
dysmenorrhea, anemia, endometrial cancer, ovarian cancer, ovarian cysts,
colorectal cancer, benign breast disease, PID, osteopenia, and osteoporosis.
Oral contraceptives may also improve cycle control, acne, and hirsutism.
3,6,8,15,24
Many clinicians believe that the
noncontraceptive benefits outweigh the risks for a majority of patients.
Regardless, the risks and benefits must be analyzed on an individual basis to
identify high-risk patients and prevent adverse events.
Summary
Since the introduction of
hormonal contraceptives to the market in 1960, there have been many changes
that provide patients with a variety of contraceptive options. This variety
allows a woman to choose a product that will be favorable to her lifestyle. It
is therefore the job of the health care provider to help women find an
agreeable contraceptive method and to ensure that each patient understands the
requirements of perfect use in order to achieve the highest possible efficacy
rates.
It is imperative that health care providers
obtain a thorough patient history upon initial selection of a contraceptive
agent in order to evaluate candidacy for hormonal contraceptives and narrow
down available products. Patients should be monitored closely after initiation
of contraceptives to identify any side effects or serious adverse events. Side
effects should be addressed as they appear, and alternative agents should be
chosen by correlating the dose and hormonal receptor affinity to the side
effects present in order to prevent contraceptive discontinuation. By
following these steps, the risks of unplanned pregnancies, side effects of
contraceptives, and product discontinuation can be minimized.
REFERENCES
1. U.S. Bureau of the Census.
Annual estimates of the population by sex and five-year age groups for the
United States: April 1, 2000 to July 1, 2003. Available at:
www.census.gov/popest/national/asrh/NC-EST2003/NC-EST2003-01.xls. Accessed
June 22, 2005.
2. Mosher WD, Martinez GM, Chandra A,
et al. Use of contraception and use of family planning services in the United
States: 1982-2002. Adv Data. 2004;350:1-36.
3. Dickerson LM, Bucci KK.
Contraception. In: DiPiro JT, Talbert RL, Yee GC, et al., eds.
Pharmacotherapy: A Pathophysiologic Approach. New York, NY: McGraw-Hill;
2002;5:1445-1461.
4. Family Health International.
Mechanisms of the contraceptive action of hormonal methods and intrauterine
devices (IUDs). Available at: www.fhi.org/en/RH/Pubs/factsheets/mechact.htm.
Accessed June 22, 2005.
5. Perlman SE, Richmond DM, Sabatini
MM, et al. Contraception: myths, facts and methods. J Reprod Med.
2001;46(2 suppl):169-177.
6. Petitti DB. Combination
estrogen-progestin oral contraceptives. N Engl J Med.
2003;349:1443-1450.
7. Family Health International. Using
progestin-only pills correctly. Available at:
www.fhi.org/en/RH/Pubs/factsheets/POcorrect.htm. Accessed June 23, 2005.
8. Edwards LA. An update on oral
contraceptive options. Formulary. 2004;39:104-121.
9. Micronor [package insert].
Raritan, NJ: Ortho-McNeil. January 2002.
10. Family Health International.
Using pills correctly. Available at:
www.fhi.org/en/RH/Pubs/factsheets/pillcorrect.htm. Accessed June 23, 2005.
11. Allen J. Hormonal contraception.
Pharmacist's Letter. 2003;19:Number 191010.
12. Gomes MP, Deitcher SR. Risk of
venous thromboembolic disease associated with hormonal contraceptives and
hormone replacement therapy: a clinical review. Arch Intern Med.
2004;164:1965-1976.
13. La Vecchia C. Oral
contraceptives, menopause hormone replacement therapy, and risk of stroke.
Maturitas. 2004;47:265-268.
14. Sidney S, Siscovick D, Petitti
DB, et al. Myocardial infarction and use of low-dose oral contraceptives: a
pooled analysis of two U.S. studies. Circulation. 1998;98:1058-1063.
15. Batur P, Elder J, Mayer M. Update
on contraception: benefits and risks of the new formulations. Cleve Clin J
Med. 2003;70(8):681-696.
16. Ortho Evra Package Insert.
Raritan, NJ: Ortho-McNeil. May 2005.
17. Potts RO, Lobo RA. Transdermal
drug delivery: clinical considerations for the obstetrician-gynecologist.
Obstet Gynecol. 2005;105(5 pt 1):953-961.
18. Wickersham RM, Novak KK, eds.
Drug Facts and Comparisons. St. Louis, MO: Wolters Kluwer Health, Inc.;
2005.
19. Mirena [package insert].
Montville, NJ: Berlex Laboratories. April 2003.
20. Herndon EJ, Zieman M. New
contraceptive options. Am Fam Physician. 2004;69:853-860.
21. NuvaRing [package insert]. West
Orange, NJ: Organon. October 2001.
22. Depo-Provera [package insert].
Kalamazoo, MI: Pharmacia & Upjohn Company. November 2004.
23. Depo-subQ Provera 104 [package
insert]. New York, NY: Pharmacia & Upjohn Company. March 2005.
24. Family Health International.
Benefits and risks of combined oral contraception. Available at:
www.fhi.org/en/RH/Pubs/factsheets/OCriskben.htm. Accessed June 23, 2005.
To comment on this article,
contact
editor@uspharmacist.com.






