US Pharm. 2018;43(8)8-11.
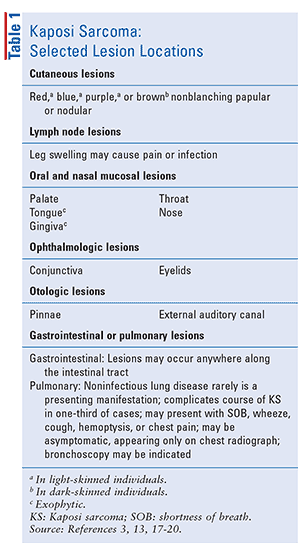
Kaposi sarcoma (KS) is a low-grade vascular tumor associated with human herpesvirus 8 infection (HHV8).1 Predominantly a cutaneous malignancy, KS is a locally aggressive endothelial tumor belonging to the family of vascular sarcoma.2 KS lesions may be confused with other vascular lesions, such as angiomas and pyogenic granulomas.3 TABLE 1 provides a list of selected locations where lesions clinically present in KS.
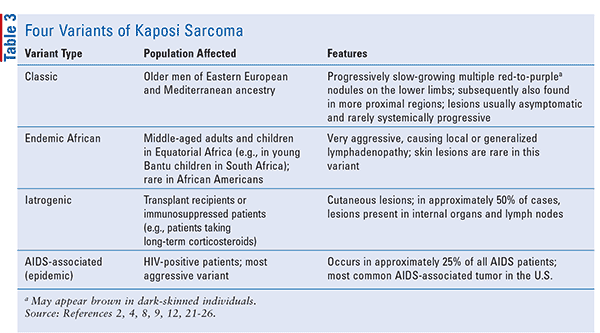
The classic KS variant is known to be more common in older men of Eastern European and Mediterranean ancestry and is characterized by a localized and indolent clinical behavior (TABLE 2).4 KS may also be discovered incidentally in older men.5

According to etiology and epidemiology, four variants have been described; TABLE 3 outlines the population affected and clinical features of each variant, or clinical subtype: classic, endemic African, iatrogenic, and AIDS-associated (epidemic). HHV8 has been isolated from all clinical subtypes of KS, which suggests that this is the etiologic factor.2 In healthy individuals, HHV8 infection generally does not cause symptoms. As with other cell-transforming human DNA viruses, infection with HHV8 alone is not sufficient for the development of KS and additional cofactors are required.6 In immunocompromised individuals, however, such as renal-transplant patients, HHV8 can potentially trigger KS; cells harboring HHV8 multiply, and the characteristic lesions form via unknown mechanisms.7 Patients with HIV have the highest risk of KS.7
KS occurs in AIDS-affected homosexual men 20 times more frequently than in nonhomosexual AIDS patients with the same degree of immunodeficiency.1 AIDS-associated KS has no preferred locations in the body but is widely scattered, and involvement of the lymph nodes and intestinal tract occurs relatively early.8,9 Further, in light of the national opioid epidemic and the risk of acquiring HIV infection from illicit drug use with sharing of needles from an HIV-infected source (a risk of 1:150), pharmacists should have a heightened awareness that KS is the most common AIDS-associated tumor in the United States.3
TREATMENT
Standard treatment for KS involves radiation therapy, surgery, chemotherapy, and biological therapy; new types of treatment are being tested in clinical trials (i.e., targeted therapy). The clinical subtype of KS will dictate the associated treatment.2 Recommendations and decisions about chemotherapy in the older adult patient must consider acceptable responses with acceptable levels of toxicity.10
Surgery, Radiation, and Chemotherapy
Surgical procedures that may be used in KS to treat small surface lesions include local excision, electrodesiccation and curettage, and cryosurgery (cryotherapy).11
In cases of classic KS, simple excision may be appropriate; these tumors are also radiosensitive. Although a less durable response is seen with the other variants of KS, similar treatments may be employed. Systemic chemotherapy (TABLE 4), however, may be warranted in more extensive disease.12 In older adults with KS lesions (e.g., oral lesions), palliative local treatment with intralesional chemotherapy or radiation is often all that is required.13 Treatment of AIDS-associated KS combines treatment for KS with treatment for AIDS: Antiretroviral therapy, which increases CD4 counts, may be effective in AIDS-related KS and should be optimized, and it can be combined with treatments such as interferon alfa (see Biological Therapy, below).11,14 In advanced AIDS-related KS, combination chemotherapy is used (e.g., daunorubicin, bleomycin, vinblastine).
With regard to the management of malignant disease in the older adult, cardiotoxicity of doxorubicin generally appears to be increased to some extent, although it depends upon the aggressiveness of the regimen.10 Clinicians should seek modalities that, in the dosages prescribed, have acceptable responses and acceptable levels of toxicity.10 Liposomal chemotherapy to treat KS uses liposomal doxorubicin to build up and slowly release the chemotherapeutic agent, rendering it more effective against sarcoma tissue and less damaging to healthy tissue.11 Of note, comorbidity generally has a negative impact on cancer-patient survival; study data in older adults indicate that hospitalization for chemotherapy-induced toxicity significantly increased with patients’ comorbidity scores.10
Biological Therapy
Biological therapy, also referred to as biotherapy or immunotherapy, involves using the patient’s immune system to fight malignancy. In vivo or in vitro substances are employed to boost, direct, or restore an individual’s natural defenses against cancer. Interferon alfa is the biologic agent used in the treatment of KS.15
Targeted Therapy
Some types of soft-tissue sarcoma have specific cellular characteristics that are vulnerable to attack via targeted drug treatments; targeted therapy has been especially helpful in gastrointestinal stromal tumors.16 Monoclonal antibody therapy (e.g., bevacizumab) and tyrosine kinase inhibitors (TKIs; e.g., imatinib mesylate) are being studied in the treatment of KS.15
Role of the Pharmacist
Pharmacists have an opportunity to educate older patients and care partners about targeted therapy by explaining in basic terms how15
• Targeted therapy identifies and attacks specific cancer cells without harming normal cells
• Monoclonal antibodies are used alone or to carry drugs, toxins, or radioactive material specifically to cancer cells, kill them, block their growth, or prevent them from spreading
• TKIs are targeted-therapy drugs that block the biological signals required for tumors to grow
Conclusion
Four variants of KS exist, with classic KS known to be more common in older men of Eastern European and Mediterranean ancestry. Pharmacists can provide education about chemotherapy and other treatment modalities, including the promise of newer biological and targeted therapies, and provide resources for them. Recommendations and decisions about chemotherapy in the older adult patient need to encompass modalities that, in the dosages prescribed, have acceptable responses with acceptable levels of toxicity. Furthermore, in light of the national opioid epidemic and the risk of acquiring HIV infection from illicit drug use with sharing of needles from an HIV-infected source, pharmacists should be aware that KS is the most common AIDS-associated tumor in the U.S.
REFERENCES
1. Arora M, Goldberg EM. Kaposi sarcoma involving the gastrointestinal tract. Gastroenterol Hepatol (NY). 2010;6(7):459-462.
2. Pearson IC, Mortimer PS. Skin problems in palliative medicine. In: Doyle D, Hanks G, Cherny N, Calman K, eds. Oxford Textbook of Palliative Medicine. 3rd ed. New York, NY: Oxford University Press; 2004:618-619.
3. Katz MH. HIV infection & AIDS. In: Papadakis MA, McPhee SJ, Rabow MW, eds. Current Medical Diagnosis & Treatment. 56th ed. New York, NY: McGraw Hill Education; 2017:1332,1338,1344.
4. Bruno V, D’Onofrio L, Frezza AM, et al. Classic Kaposi sarcoma: to treat or not to treat? BMC Res Notes. 2015;8:138.
5. Zagaria MA. Kaposi’s sarcoma: prognosis varies with form. US Pharm. 2010;35(4):20-23.
6. Iscovich J, Boffetta P, Franceschi S, et al. Classic Kaposi sarcoma: epidemiology and risk factors. Cancer. 2000;88(3):500-517.
7. Mayoclinic.com. Kaposi’s sarcoma. www.mayoclinic.org/diseases-conditions/kaposis-sarcoma/cdc-20387726. Accessed July 2, 2018.
8. Biggar RJ, Horm J, Fraumeni JF Jr, et al. Incidence of Kaposi’s sarcoma and mycosis fungoides in the United States including Puerto Rico, 1973-1981. J Natl Cancer Inst. 1984;73:89-94.
9. Beral V, Peterman TA, Berkelman RL, Jaffe HW. Kaposi’s sarcoma among persons with AIDS: a sexually transmitted infection? Lancet. 1990;335:123-128.
10. Rao AV, Cohen HJ. Oncology and aging: general principles. In: Halter JB, Ouslander JG, Tinetti ME, et al, eds. Hazzard’s Geriatric Medicine and Gerontology. 6th ed. New York, NY: McGraw Hill; 2009:1123-1135.
11. Medline Plus. Kaposi sarcoma. U.S. National Library of Medicine. Updated June 6, 2018. https://medlineplus.gov/kaposisarcoma.html#. Accessed July 12, 2018.
12. Ross RK, Casagrande JT, Dworsky RL, et al. Kaposi’s sarcoma in Los Angeles, California. J Natl Cancer Inst. 1985;75:1011-1015.
13. Shinkai K, Fox LP. Dermatologic disorders. In: Papadakis MA, McPhee SJ, Rabow MW, eds. Current Medical Diagnosis & Treatment. 56th ed. New York, NY: McGraw Hill Education; 2017:146.
14. Schwartz RA, Cohen PJ. Kaposi’s sarcoma. In: Newcomer VD, Young EM Jr, eds. Geriatric Dermatology: Clinical Diagnosis and Practical Therapy. New York, NY: Igaku-Shoin; 1989:645-652.
15. National Cancer Institute. Kaposi sarcoma treatment (PDQ®)–patient version. Updated February 14, 2018. www.cancer.gov/types/soft-tissue-sarcoma/patient/kaposi-treatment-pdq#section/_50. Accessed July 12, 2018.
16. Mayoclinic.com. Soft-tissue sarcoma. www.mayoclinic.org/diseases-conditions/soft-tissue-sarcoma/symptoms-causes/syc-20377725. Accessed July 2, 2018.
17. Riordan-Eva P. Disorders of the eyes and lids. In: Papadakis MA, McPhee SJ, Rabow MW, eds. Current Medical Diagnosis & Treatment. 56th ed. New York, NY: McGraw Hill Education; 2017:192.
18. Lustig LR, Schindler JS. Ear, nose & throat disorders. In: Papadakis MA, McPhee SJ, Rabow MW, eds. Current Medical Diagnosis & Treatment. 56th ed. New York, NY: McGraw Hill Education; 2017:214.
19. Ortel B, Bolotin D. Cancer of the skin: vascular malignancies. In: Bope ET, Kellerman RE, eds. Conn’s Current Treatment 2017. Philadelphia, PA; Elsevier:2017;904.
20. Cornett PA, Dea TO. Cancer. In: Papadakis MA, McPhee SJ, Rabow MW, eds. Current Medical Diagnosis & Treatment. 56th ed. New York, NY: McGraw Hill Education; 2017:1625,1636.
21. Laor Y, Schwartz RA. Epidemiologic aspects of American Kaposi’s sarcoma. J Surg Oncol. 1979;12:299-303.
22. Dezube BJ, Pantanowitz L, Aboulafa DM. Management of AIDS-related Kaposi sarcoma: advances in target discovery and treatment. AIDS Read. 2004;14:236-238,243-244,251-253.
23. Friedman SL, Wright TL, Altman DF. Gastrointestinal Kaposi’s sarcoma in patients with acquired immune deficiency syndrome. Endoscopic and autopsy findings. Gastroenterology. 1985;89:102-108.
24. Farge D, Lebbe C, Marjanovic Z, et al. Human herpes virus-8 and other risk factors for Kaposi’s sarcoma in kidney transplant recipients. Groupe Cooperatif de Transplantation d’Ile de France (GCIF). Transplantation. 1999;67:1236-1242.
25. Margolius L, Stein M, Spencer D, Bezwoda WR. Kaposi’s sarcoma in renal transplant recipients. Experience at Johannesburg Hospital, 1966–1989. S Afr Med J. 1994;84:16-17.
26. Shepherd FA, Maher E, Cardella C, et al. Treatment of Kaposi’s sarcoma after solid organ transplantation. J Clin Oncol. 1997;15:2371-2377.
27. Epocrates.com. Epocrates Plus Version 15.12.1. Updated July 16, 2018.
To comment on this article, contact rdavidson@uspharmacist.com.