US Pharm. 2013;38(10):44-48.
ABSTRACT: Rheumatoid arthritis (RA), a debilitating autoimmune disease, is three times more common in women and occurs most frequently in middle age. Joint swelling and deformity, autoantibody production, muscle stiffness and pain, and many systemic manifestations characterize RA. Diagnosis is based on physical examination and the patient’s history of joint pain. Despite major advances in treatment, the main etiology of RA remains unknown. Rheumatoid factor and anticyclic citrullinated peptide antibodies have been incorporated into clinical practice as disease biomarkers. RA treatment involves patient education and the use of nonsteroidal anti-inflammatory drugs and disease-modifying antirheumatic drugs. Newer drugs, such as golimumab, certolizumab, and tocilizumab, have been added to the list of biologic agents used to manage RA.
Rheumatoid arthritis (RA) is an autoimmune disease that is associated with progressive disability, systemic complications, early death, and socioeconomic costs.1 The chronic systemic inflammatory response associated with RA affects most joints and their surrounding tissues. RA can occur at any age, but is more common in middle age. RA that may be attributed to hormonal changes affects more women than men. In North America and Northern Europe, estimates of RA incidence range from 20 to 50 cases per 100,000 population, with a prevalence of 0.5% to 1.1% per 100 population.2
CHARACTERISTICS AND DIAGNOSIS
Despite extensive research, the main etiology of RA remains unknown. However, some contributory factors have been suggested, including genetics, infectious agents, oral contraceptives, and smoking.3 Most studies have indicated that genetics plays a major role in the development of RA. More than 80% of patients carry the epitope of the HLA-DRB1*04 cluster, and patients expressing two HLA-DRB1*04 alleles are at greater risk for nodular disease, major organ involvement, and surgery related to joint destruction.4
RA is characterized by synovial inflammation and hyperplasia; autoantibody production (rheumatoid factor [RF] and anticitrullinated protein antibody); cartilage and bone destruction; and systemic features, including cardiovascular, pulmonary, psychological, and skeletal disorders.5 The clinical hallmark of RA is polyarticular synovial inflammation of peripheral joints, typically in the hands (metacarpophalangeal joints and proximal interphalangeal joints), which results in pain, stiffness, and often some degree of irreversible joint damage, deformity, and disability. Additionally, a significant systemic inflammatory state exists that may promote various other extra-articular effects, including coronary artery disease, pulmonary fibrosis, osteoporosis, and vasculitis.6
Clinical diagnosis is based on the patient’s history of joint pain and stiffness and a physical examination for symmetric polyarticular joint swelling. Laboratory tests, including radiographs and blood tests, can supply useful information confirming or contributing to the diagnosis of RA. Many autoantibodies have been reported in the blood of RA patients, but only RF and anticyclic citrullinated peptide antibodies have been incorporated into routine clinical practice.7
NONPHARMACOLOGIC TREATMENT
The treatment of RA begins with educating patients, their families, and their caregivers. Patients need to be aware that proper diet and exercise, sufficient rest, and weight reduction are just as important as adherence to drug therapy in managing the disease. Once patients understand the nature of RA and its potential course, emotional support should be provided, since the risk of depression is two to three times greater in RA patients than in patients with other chronic, debilitating diseases.8
Research has shown that dietary changes may confer a benefit in managing RA symptoms. Patients are encouraged to follow a Mediterranean-style diet (high intake of n-3 polyunsaturated fatty acids, such as deep-sea fish and nuts, and low intake of saturated fats, such as red meat) to reduce inflammation and joint pain and boost the immune system. Iron and folic acid supplementation are also important, since many RA patients experience anemia.9
Occupational and physical therapy, in conjunction with passive exercise (e.g., range-of-motion exercises) and rest, are paramount in the nonpharmacologic management of RA. Assistive devices (canes, crutches, braces, reachers, etc.) can provide substantial symptomatic relief for patients with mild-to-moderate symptoms. In patients with moderate-to-severe disease, synovectomy (removal of proliferated synovium) and other surgical procedures frequently are warranted.8
PHARMACOTHERAPY
Pharmacotherapy for RA involves the use of nonsteroidal anti-inflammatory drugs (NSAIDs) and disease-modifying antirheumatic drugs (DMARDs). See TABLE 1 for a summary.
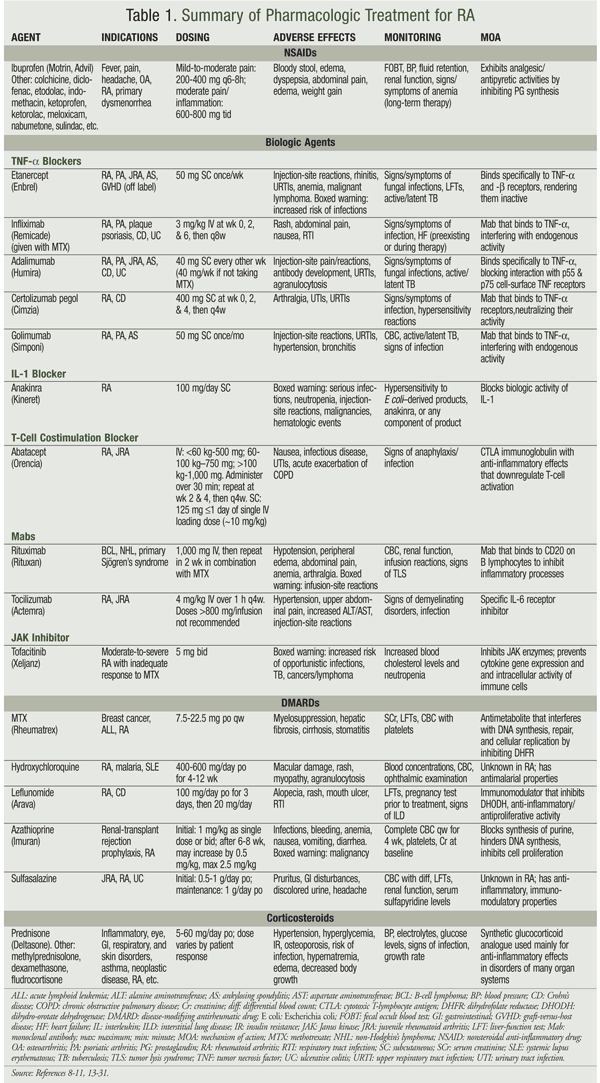
NSAIDs
NSAIDs work by inhibiting prostaglandin synthesis, from which they derive their anti-inflammatory and analgesic properties. They do not slow disease progression and are incapable of preventing joint deformity; therefore, they are viewed as adjuncts to DMARD treatment and are not recommended as monotherapy for RA.10,11 Salicylates (aspirin and nonaspirin) and cyclo-oxygenase-2 inhibitors are equally efficacious when used at high anti-inflammatory doses. Common adverse effects (AEs) of these agents are nausea, dyspepsia, gastrointestinal (GI) ulceration or hemorrhage, and renal dysfunction.12 It is important to assess the patient’s specific risk factors for GI ulceration and renal dysfunction prior to NSAID initiation.
DMARDs
The American College of Rheumatology (ACR) recommends the use of DMARDs as first-line agents for the treatment of RA, and practice guidelines instruct that DMARDs be initiated within the first 3 months of symptom onset for more favorable outcome and decreased mortality.10,11 Cyclophosphamide, cyclosporine, D-penicillamine, glucocorticoids, gold, and minocycline are older DMARDs that are less frequently used because of high toxicity or decreased efficacy.10 Hydroxychloroquine, leflunomide (LEF), methotrexate (MTX), minocycline, and sulfasalazine are first-line agents, and a combination of two or three of these drugs may be used when appropriate. Recommended combinations include MTX plus hydroxychloroquine, MTX plus LEF, and MTX plus sulfasalazine.8,11
LEF (Arava): This drug is an immunomodulating agent that works by inhibiting dihydro-orotate dehydrogenase.12 A loading dose of 100 mg orally once daily for 3 days followed by a maintenance dose of 20 mg daily is recommended, but this can be decreased to 10 mg daily for increased tolerability. Because of LEF’s long half-life, the loading dose is recommended for achieving steady state more quickly. There is a boxed warning to avoid LEF in pregnancy. Monitoring of serum alanine transferase is recommended monthly for the first 6 months of LEF treatment, and every 6 to 8 weeks thereafter. The comparative efficacy and safety of MTX or LEF in combination with rituximab (RTX) were examined in a retrospective study.13 No significant difference was found between the RTX + MTX group and the RTX + LEF group when evolution of disease activity and functional disability and AEs were compared.
Azathioprine (Imuran): Azathioprine (AZA) is an imidazolyl derivative of 6-mercaptopurine that acts like an immunosuppressant, although the exact mechanism is unknown.14 It is approved for the treatment of RA and for renal transplant rejection prophylaxis. The efficacy of MTX and AZA were compared in a follow-up study involving treatment for up to 4 years.15 More patients in the AZA arm switched from the MTX arm, and measures of clinical improvement were more pronounced in the MTX arm.
MTX (Rheumatrex): This agent is an antimetabolite that interferes with DNA synthesis, repair, and replication in malignant cells. It is currently the DMARD of choice for initiation in patients with RA.14 Although intramuscular administration is an option, MTX is generally administered orally starting at 7.5 mg once weekly or 2.5 mg every 12 hours for three doses per week (pulse dosing).
Common AEs of MTX therapy are GI upset (nausea, vomiting, and diarrhea), stomatitis, malaise, dizziness, and mild alopecia.15 Less common, more severe AEs include dose-related bone marrow suppression, liver toxicity, infections, rheumatoid nodules, leukopenia, pneumonitis, thrombocytopenia, and renal failure. MTX is contraindicated in pregnancy, preexisting blood dyscrasias, AIDS, alcoholic liver disease, severe hepatic or renal dysfunction, and hypersensitivity to the drug or any of its components. Caution should be exercised in women of childbearing potential or in patients with peptic ulcer or ulcerative colitis.
Providers and patients should be mindful of the major drug interactions for MTX.15 MTX is protein bound and excreted renally; therefore, salicylates, probenecid, penicillin, and ciprofloxacin may be problematic. Trimethoprim may increase the risk of myelosuppression when taken with MTX, and NSAIDs increase serum concentrations and risk of MTX toxicity when the two agents are coadministered.
BIOLOGICS
Biologics are generally indicated when the prognosis is poor or when there is a poor response to nonbiologic DMARDs. These medications are administered either IV or subcutaneously (SC) and may be used in combination with MTX.
Tumor Necrosis Factor (TNF)–Alpha Blockers
Etanercept (Enbrel): Approved in 1998, etanercept was the first drug in this class. It is administered 50 mg weekly SC. It has a boxed warning for increased risk of infections, including tuberculosis (TB) and bacterial and fungal infections.16 All patients should have a negative skin test for TB prior to initiating etanercept, and live vaccines are contraindicated in patients taking etanercept. Etanercept carries a boxed warning for lymphomas, which have occurred in children and adolescents using this medication although the drug is not approved for use in pediatric patients. This Pregnancy Category B drug has a pregnancy registry that monitors outcomes in pregnant women. A medication guide is required to be dispensed with etanercept. The drug must be stored in a refrigerator and is available in pen and vial forms.
A randomized, double-blind, placebo-controlled trial compared etanercept 10 mg or 25 mg twice weekly with MTX once weekly (average dose 19 mg/week).17 Etanercept 25 mg was found to be more effective than etanercept 10 mg and MTX in improving response rates (RR) and progression of joint erosion, with more MTX patients having AEs.18
Infliximab (Remicade): This agent, which was approved in 1998, must be taken in conjunction with MTX.19 The dosage for RA is 3 mg/kg at 0, 2, and 6 weeks and every 8 weeks thereafter; it may be titrated up to 10 mg/kg or given every 4 weeks. Because infliximab is administered IV over at least 2 hours, it cannot be self-administered. Infliximab has a boxed warning for the risk of infections and lymphomas, and the rate of granulomatous infections after infliximab treatment was three times greater than that for etanercept.20
Adalimumab (Humira): Approved in 2002, adalimumab is dosed at 40 mg SC every other week for RA.21 It has a boxed warning for increased risk of infections and lymphomas and is a Pregnancy Category B drug that has a pregnancy registry. Like etanercept, adalimumab must be stored in the refrigerator and is available both as a pen and as prefilled syringes. In one trial, inadequate responders to MTX were randomized to MTX plus adalimumab 40 mg every other week, adalimumab 20 mg every other week, or placebo.22 At 52 weeks, all patients were switched to 40 mg adalimumab every other week and followed for an additional 5 years. The 52-week delay in adding adalimumab to the placebo arm resulted in increased radiographic progression and less clinical remission at year 5.
Interleukin (IL)-1 Blocker
Anakinra (Kineret): Anakinra is the recombinant form of nonglycosylated human IL-1 receptor antagonist expressed in Escherichia coli. It binds competitively to both type I and type II IL-1 receptors.23 Anakinra is administered SC 100 mg daily, at the same time every day. In a randomized, double-blind, placebo-controlled trial of anakinra versus placebo in patients already receiving stable doses of MTX, there was a 46% RR in the anakinra group versus a 19% RR in the placebo group, using ACR20 response criteria (20% improvement) as a measure.24
Selective T-Cell Costimulation Modulator
Abatacept (Orencia): This agent, which is infused IV, utilizes weight-based dosing and requires a loading dose. Subsequent infusions should be administered at 2 weeks and 4 weeks after the loading dose, and every 4 weeks thereafter. A study compared the efficacy and safety of abatacept or infliximab versus placebo in patients with an inadequate response to MTX.25 Both abatacept and infliximab were associated with statistically significant changes in disease activity (based on erythrocyte sedimentation rate) versus placebo. Compared with infliximab, abatacept had a lower incidence of AEs, serious AEs, and serious infections, and had similar efficacy.
Human Monoclonal Antibodies (Mabs)
Rituximab (Rituxan): Rituximab is a CD20-directed cytolytic antibody indicated for the treatment of RA in combination with MTX in patients with an inadequate response to one or more TNF antagonists.26 It is infused IV and is dosed according to body weight. Patients should be premedicated with a glucocorticoid, an antihistamine, and acetaminophen 30 minutes prior to infusion. A comparison of the addition of rituximab or placebo in patients with an inadequate response to MTX found significantly improved RRs in the rituximab group compared with the placebo group.27
Tocilizumab (Actemra): This humanized Mab inhibits IL-6 receptor signaling by binding to both the soluble and membrane-bound IL-6 receptors.28 Tocilizumab is initiated as a 4 mg/kg IV infusion over 1 hour every 4 weeks, and the dose is increased to 8 mg/kg every 4 weeks (maximum 800 mg/infusion) based on clinical response. A study evaluated the efficacy of tocilizumab monotherapy versus MTX monotherapy in patients who had not previously failed other biologic agents or MTX. Tocilizumab was superior to MTX, with 69.9% of tocilizumab patients meeting ACR20 response criteria in 6 weeks versus 52.5% of MTX patients.29
Certolizumab (Cimzia): This agent, an Mab directed against TNF-alpha, is indicated for the treatment of moderate-to-severe RA and Crohn’s disease. Certolizumab is administered SC at a dose of 400 mg at weeks 0, 2, and 4, then 200 mg every other week or 400 mg every 4 weeks.30 In the phase III RAPID 2 trial, patients with active adult-onset RA were randomized to MTX plus certolizumab 400 mg at weeks 0, 2, and 4 followed by 200 mg or 400 mg, or to MTX plus placebo, every 2 weeks for 24 weeks. The ACR20 RR was 57.3% in the 200-mg arm, 57.6% in the 400-mg arm, and 8.7% in the placebo arm. Certolizumab 400 mg did not offer a significant advantage over 200 mg, and both dosages were significantly better than placebo.31
New Agents
Tofacitinib (Xeljanz): Tofacitinib, a Janus kinase inhibitor, was approved in November 2012 for the treatment of moderate-to-severe RA.32 It should not be used in combination with other biologics, but may be used with MTX or other nonbiologic DMARDs. It is an oral tablet taken 5 mg twice daily. Tofacitinib carries a boxed warning regarding infections and lymphomas. In a 12-month, phase III trial, tofacitinib 5 mg twice daily, tofacitinib 10 mg twice daily, and adalimumab 40 mg once every 2 weeks were compared with placebo.33 All patients were receiving stable doses of MTX. The RR (defined as meeting ACR20 response criteria) was higher in the tofacitinib 5 mg and 10 mg groups (51.5% and 52.6%, respectively) than in the adalimumab and placebo groups (47.2% and 28.3%, respectively). Tofacitinib was concluded to be significantly superior to placebo, with efficacy similar to that of adalimumab.
Golimumab (Simponi): Golimumab is the first approved once-monthly SC anti-TNF agent, with adult patients receiving an injection of 50 mg. Golimumab must be administered with MTX. A double-blind, placebo-controlled, phase III study evaluated the efficacy of golimumab 2 mg/kg plus MTX in patients with active RA who were receiving MTX. At week 14, a significantly larger proportion of golimumab-plus-MTX patients than placebo-plus-MTX patients achieved ACR20 response (59% vs. 25%), a disease-activity score of good/moderate response (81% vs. 40%), and greater median improvement in health-assessment questionnaire scores (0.500 vs. 0.125).34 Prior to treatment initiation, patients should be current with pneumococcal, influenza, hepatitis, human papillomavirus, and herpes zoster vaccinations, in addition to being screened for TB, owing to the propensity of these drugs to cause serious infections.35
CONCLUSION
RA is a debilitating disease that affects many people in the United States. Advances in treatment that involve newer drug agents, along with the therapeutic issues arising from older regimens, render patient and provider education extremely important in the quest to achieve optimal patient outcomes. Pharmacists should be well prepared to discuss the common concerns of patients and their caregivers upon initiation of DMARDs and biologic agents, which are currently the mainstays of RA treatment. When they are empowered with knowledge of treatment options for RA and receiving the support of their health care providers, patients are better able to actively participate in the management of their disease.
REFERENCES
1. McInnes IB, Schett G. Cytokines in the pathogenesis of rheumatoid arthritis. Nat Rev Immunol. 2007;7:429-442.
2. Tobón GJ, Youinou P, Saraux A. The environment, geo-epidemiology, and autoimmune disease: rheumatoid arthritis. J Autoimmun. 2010;35:10-14.
3. Gabriel SE. The epidemiology of rheumatoid arthritis. Rheum Dis Clin North Am. 2001;27:269-281.
4. Smolen JS, Aletaha D, Koeller M, et al. New therapies for treatment of rheumatoid arthritis. Lancet. 2007;370:1861-1874.
5. McInnes IB, Schett G. The pathogenesis of rheumatoid arthritis. N Engl J Med. 2011;365:2205-2219.
6. Smith HS, Smith AR, Seidner P. Painful rheumatoid arthritis. Pain Physician. 2011;14:E427-E458.
7. Farheen K, Agarwal SK. Assessment of disease activity and treatment outcomes in rheumatoid arthritis. J Manag Care Pharm. 2011;17(9 suppl B):S9-S13.
8. Koda-Kimble MA, Young LY, Alldredge BK, et al, eds. Applied Therapeutics: The Clinical Use of Drugs. 9th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2009:43-1–43-4.
9. Wall R, Ross RP, Fitzgerald GF, Stanton C. Fatty acids from fish:
the anti-inflammatory potential of long-chain omega-3 fatty acids. Nutr Rev. 2010;68:280-289.
10. Wells BG, DiPiro JT, Schwinghammer TL, DiPiro C, eds. Pharmacotherapy Handbook. 8th ed. New York, NY: McGraw-Hill Medical; 2012:469-487.
11. American College of Rheumatology Subcommittee on Rheumatoid
Arthritis Guidelines. Guidelines for the management of rheumatoid
arthritis: 2002 update. Arthritis Rheum. 2002;46:328-346.
12. Arava (leflunomide) product information. Bridgewater, NJ: Sanofi-aventis U.S. LLC; November 2012.
13. Narváez J, Diaz-Torné C, Ruiz JM, et al. Comparative
effectiveness of rituximab in combination with either methotrexate or
leflunomide in the treatment of rheumatoid arthritis. Semin Arthritis Rheum. 2011;41:401-405.
14. Arthritis Foundation. Focus on rheumatoid arthritis.
www.arthritistoday.org/conditions/rheumatoid-arthritis. Accessed
February 12, 2013.
15. Rheumatrex (methotrexate) product information. Fort Lee, NJ: DAVA Pharmaceuticals, Inc; July 2009.
16. Schuna AA. Rheumatoid arthritis. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 8th ed. New York, NY: McGraw-Hill Medical; 2011.
17. Enbrel (etanercept) product information. Thousand Oaks, CA: Immunex Corp; December 2012.
18. Bathon JM, Martin RW, Fleischmann RM, et al. A comparison of
etanercept and methotrexate in patients with early rheumatoid arthritis.
N Engl J Med. 2000;343:1586-1593.
19. Remicade (infliximab) product information. Horsham, PA: Janssen Biotech, Inc; October 2011.
20. Wallis RS, Broder MS, Wong JY, et al. Granulomatous infectious diseases associated with tumor necrosis factor antagonists. Clin Infect Dis. 2004;38:1261-1265.
21. Humira (adalimumab) product information. North Chicago, IL: Abbott Laboratories; September 2012.
22. Keystone EC, Kavanaugh A, Weinblatt ME, et al. Clinical
consequences of delayed addition of adalimumab to methotrexate therapy
over 5 years in patients with rheumatoid arthritis. J Rheumatol. 2011;38:855-862.
23. Anakinra. DRUGDEX System [online database]. Greenwood Village, CO: Thomson Reuters (Healthcare) Inc; 2012.
24. Cohen S, Hurd E, Cush J, et al. Treatment of rheumatoid arthritis
with anakinra, a recombinant human interleukin-1 receptor antagonist,
in combination with methotrexate: results of a twenty-four-week,
multicenter, randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 2002;46:614-624.
25. Schiff M, Keiserman M, Codding C, et al. Efficacy and safety of
abatacept or infliximab vs placebo in ATTEST: a phase III, multi-centre,
randomised, double-blind, placebo-controlled study in patients with
rheumatoid arthritis and an inadequate response to methotrexate. Ann Rheum Dis. 2008;67:1096-1103.
26. Rituxan (rituximab) product information. South San Francisco, CA: Genentech, Inc; October 2012.
27. Emery P, Deodhar A, Rigby WF, et al. Efficacy and safety of
different doses and retreatment of rituximab: a randomised,
placebo-controlled trial in patients who are biological naïve with
active rheumatoid arthritis and an inadequate response to methotrexate
(Study Evaluating Rituximab’s Efficacy in MTX iNadequate rEsponders
(SERENE)). Ann Rheum Dis. 2010;69:1629-1635.
28. Actemra (tocilizumab) product information. South San Francisco, CA: Genentech, Inc; October 2012.
29. Jones G, Sebba A, Gu J, et al. Comparison of tocilizumab
monotherapy versus methotrexate monotherapy in patients with moderate to
severe rheumatoid arthritis: the AMBITION study. Ann Rheum Dis. 2010;69:88-96.
30. Cimzia (certolizumab) product information. Smyrna, GA: UCB, Inc; November 2012.
31. Smolen J, Landewé RB, Mease P, et al. Efficacy and safety of
certolizumab pegol plus methotrexate in active rheumatoid arthritis: the
RAPID 2 study. A randomized controlled trial. Ann Rheum Dis. 2009;68:797-804.
32. Xeljanz (tofacitinib) product information. New York, NY: Pfizer Labs; November 2012.
33. van Vollenhoven RF, Fleischmann R, Cohen S, et al. Tofacitinib or adalimumab versus placebo in rheumatoid arthritis. N Engl J Med. 2012;367:508-519.
34. Weinblatt ME, Bingham CO III, Mendelsohn AM, et al. Intravenous
golimumab is effective in patients with active rheumatoid arthritis
despite methotrexate therapy with responses as early as week 2: results
of the phase 3, randomised, multicentre, double-blind,
placebo-controlled GO-FURTHER trial. Ann Rheum Dis. 2013;72:381-389.
35. Singh J, Furst DE, Bharat A, et al. 2012 update of the 2008
American College of Rheumatology recommendations for the use of
disease-modifying antirheumatic drugs and biologic agents in the
treatment of rheumatoid arthritis. Arthritis Care Res (Hoboken). 2012;64:625-639.
To comment on this article, contact rdavidson@uspharmacist.com.






