US Pharm. 2011;36(4):46-52.
In the United States, diabetic retinopathy (DR) affects approximately 86% of patients with type 1 diabetes and 40% with type 2 diabetes. In one study, 97% of type 1 cohorts eventually developed DR,1 the primary cause of blindness in adult Americans.2 The informed pharmacist can assist patients with preserving their eyesight by modifying their risk for this progressive eye disease.
Classification
Diabetic retinopathy is broadly classified according to disease progression. The initial stage, nonproliferative DR (NPDR; previously termed “background” retinopathy), can be further categorized as mild, moderate, or severe. Proliferative DR (PDR) represents the advanced stage of the disease.3
Anatomy and Physiology
The human eye is divided into anterior and posterior segments (FIGURE 1A). The anterior segment includes the cornea, pupil, iris, lens, and the aqueous humor. The posterior segment includes the vitreous humor, choroid, sclera, macula, optic nerve, and retina.
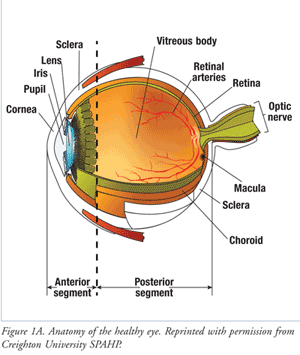
The wall of the eye has three layers. The outermost white layer is the sclera, a tough, fibrous tissue that protects the eye and helps maintain its shape. The cornea, a domed, transparent structure, is continuous with the sclera and covers the anterior eye. The vascular middle layer, or uvea, consists of the choroid in the posterior segment and the iris in the anterior segment. The retina, or inner layer, lines the back two-thirds of the choroid and possesses a rich vascular supply.
Light passes through the pupil and enters the posterior segment of the eye via the crystalline lens. Fluid fills the entire eye, supporting its structure and various metabolic functions. The anterior segment contains the clear, lymph-like, watery aqueous humor, while the posterior segment holds clear, jellylike vitreous humor. Unlike the retina, a healthy vitreous chamber contains no blood vessels.
As light enters the eye, it is bent by the cornea and lens, passes through the vitreous, and is focused on the retina. Retinal neural tissues transform light into electrical impulses, which are transmitted by the optic nerve to the brain and interpreted as visual images. The macula is a small area just lateral to the center of the retina that provides detailed central visual acuity.
Pathophysiology of Diabetic Retinopathy
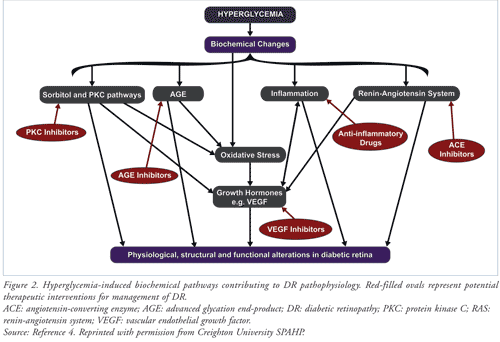
Although clinical manifestations of DR occur after several years, biochemical, molecular, and hemodynamic changes begin in the early stages of diabetes. Chronic intracellular hyperglycemia, consequent to an absolute or incremental insulin deficit, triggers a cascade of biochemical mechanisms at the cellular level that drive the disease process. These include activation of the aldose reductase (sorbitol) and protein kinase C (PKC) pathways and the renin-angiotensin system (RAS), oxidative stress (OS), accumulation of advanced glycation end-products (AGEs), and release of inflammatory mediators and vascular endothelial growth factor (VEGF) that damage ophthalmic vascular endothelial cells. Ultimately, microvascular damage and retinal dysfunction occur (FIGURE 2).4 These biochemical mechanisms are potential targets for novel therapeutic interventions.
Clinical Findings
The earliest clinical stage of DR, mild NPDR, is characterized by the presence of tiny swellings or microaneurysms in the retinal capillary walls. With progression to moderate NPDR, fluid that contains lipids seeps from leaky damaged capillaries to eventually form lipid-rich deposits known as hard exudates. Accumulation of fluid in retinal tissues results in edema. As capillary damage intensifies, blood escapes into retinal tissues (retinal hemorrhages). In some cases, minor hemorrhages may occur during mild NPDR. Where the body attempts to repair vessel damage, capillary occlusions may occur, and ischemic retina develops due to oxygen deficits distal to the blockage. These ischemic areas appear as “cotton wool” spots on a retinal exam. At this stage, there is generally no evidence of visual loss, although some patients may experience blurriness in central or peripheral vision. Severe NPDR is marked by severe hemorrhages, microaneurysms, and exudate formation, and requires medical attention (FIGURE 1B).3
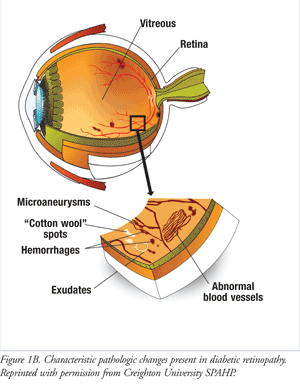
Retinopathy within the macular area of the retina marks the onset of diabetic macular edema (DME) or diabetic macular ischemia. DME is the most common cause of vision loss in DR.5 While loss of central vision may range from mild to severe, peripheral vision is sustained in DME. Moreover, DME may occur at any stage in the disease process.
PDR, the advanced stage of the disease, is typified by occlusion of blood vessels with ensuing ischemia. The retina attempts to replenish its oxygen supply by upregulating growth factors, such as VEGF, which stimulate new blood vessel formation (neovascularization) (FIGURE 1B). The new blood vessels may extend into the vitreous and are structurally and functionally abnormal. Their thin fragile walls rupture easily, allowing bleeding into the vitreous humor and development of scar tissue on damaged blood vessels (fibrovascular damage). Fibrovascular tissue may pull on the retina and ultimately result in retinal detachment, which patients experience as spots floating in the visual field, light flashes, or severe vision loss. PDR may cause more severe vision loss than NPDR and can affect both central and peripheral vision. (Refer to FIGURE 3 for photos representative of normal vision and DR.) If neovascularization develops in the iris and anterior chamber tissues, neovascular glaucoma may result. Both this disorder and retinal detachment are considered medical emergencies that may cause intractable blindness without immediate medical attention.3

Risk Factors
The major risk factors associated with developing DR are duration of diabetes and severity of hyperglycemia.3 Once retinopathy is established, hyperglycemic severity is more closely associated with disease progression.1,6,7 Acceleration of DR has been linked to hypertension in diabetic patients.3 As diabetes duration is nonmodifiable, attention is focused on the two important modifiable risk factors for DR—hyperglycemia and hypertension.1,5-7 Patients with type 1 diabetes are at increased risk for DR during puberty and pregnancy, while dyslipidemia, race, and genetics have a more tenuous link to DR.1,6,7
Goals of Therapy
The ultimate therapeutic objective in DR management is to prevent, slow, or reverse vision loss in order to maintain or improve vision-related quality of life (QOL).3 Specific goals include:
- Identify patients at risk for DR
- Promote partnership of patient and primary care provider to optimally manage systemic disorders, especially hyperglycemia and hypertension
- Encourage lifelong evaluation of retinopathy progression
- Treat patients at risk for DR-related vision loss
- Minimize therapeutic side effects that may adversely affect vision or QOL
- Refer patients with DR-related vision impairment to visual rehabilitation services.
Pharmacists must recognize that glycemic control remains the most effective prophylactic approach to managing DR. The Diabetes Control and Complications Trial (DCCT) showed that glucose maintained in the near-normal range reduced the risk of DR onset by 76% and disease progression by 54%.8 Hemoglobin A1C (A1C) £7% correlates to a decreased risk of both initial development and progression to more serious forms of DR.1,6 While more rigorous glycemic goals may modestly decrease risks related to DR, the morbidity and mortality associated with hypoglycemia are thought to outweigh potential benefits.1,6 Because good A1C control does not eliminate the risk for developing DR, all patients with diabetes should be regularly screened for pathologic changes.
The American Diabetes Association (ADA) recommends blood pressure <130/80 mmHg to decrease the risk of developing DR and to slow its progression.6 Hypertension should be closely monitored, as each elevation of 10 mmHg (systolic) may result in a 10% increased risk of developing DR and a 15% greater chance of progression to PDR.1 However, the Action to Control Cardiovascular Risk in Diabetes (ACCORD) Eye Study failed to show a decreased rate of DR progression with intensive blood pressure lowering (<120 mmHg) in type 2 diabetes.9 Further studies are needed to elucidate whether lowering blood pressure beyond a threshold level provides any beneficial effect on DR progression.5
Treatment Strategies
Because DR is asymptomatic in the early stages, patients with diabetes require regular comprehensive eye examinations to facilitate early interventions that may minimize disease progression. A dilated eye examination should be performed, as well as measurement of visual acuity and intraocular pressure (IOP). A fluorescein angiogram to assess retinal vasculature is recommended if DME is suspected.3
The most important visual complications in DR are PDR and DME. For the past several decades, surgical treatment has been the standard therapy for both complications. However, a few drug classes are under evaluation for pharmacologic intervention in DR. A combination of surgical and pharmacologic therapies may be necessary to achieve treatment goals.3
Surgical Management of DR
Focal Laser Photocoagulation (FLP): This procedure, which is utilized in DME management, involves the use of a laser beam to seal leaky blood vessels, thereby preserving normal retinal thickness and function. Although it does not restore vision, FLP may attenuate vision loss. Adverse effects include an initial decrease in central vision. Inadvertent central retinal burns may lead to permanent central blindness.3
Panretinal (Scatter) Laser Photocoagulation (PLP): PLP is aimed at arresting leakage and neovascularization of blood vessels, thereby reducing the risk of vitreous hemorrhage and retinal detachment. Since PLP involves application of laser light in the peripheral retina, some patients may experience compromised peripheral vision. Loss of accommodation and central vision have been reported in some patients. PLP is the mainstay of PDR and severe NPDR therapy.3
Vitrectomy: This procedure involves removal of vitreous humor and may be warranted in severe PDR to attenuate vision loss secondary to vitreous hemorrhage and retinal detachment involving the macula.7
Pharmacologic Management of DR
Although effective in arresting the progression of DR, surgical photocoagulation procedures destroy retinal tissue and do not improve vision. Therefore, the development of noninvasive treatment strategies is warranted. Intravitreal delivery (into the vitreous chamber) of corticosteroids and VEGF inhibitors may soon become a popular adjunctive measure, or an alternative to photocoagulation, for management of DR complications.
Corticosteroids: These agents possess anti-inflammatory properties and the ability to downregulate VEGF, thereby reducing leakage from damaged retinal capillaries. Intravitreal triamcinolone acetonide (IVTA) is currently used as an off-label adjunctive treatment for DME and may be useful in PDR.10 A biodegradable, extended-release dexamethasone implant has shown promise for DME management, with a potentially superior side-effect profile compared to IVTA.11 Corticosteroids should be used cautiously in patients with a history of cataracts and glaucoma. A summary of dosing and adverse effects is provided in TABLE 1.
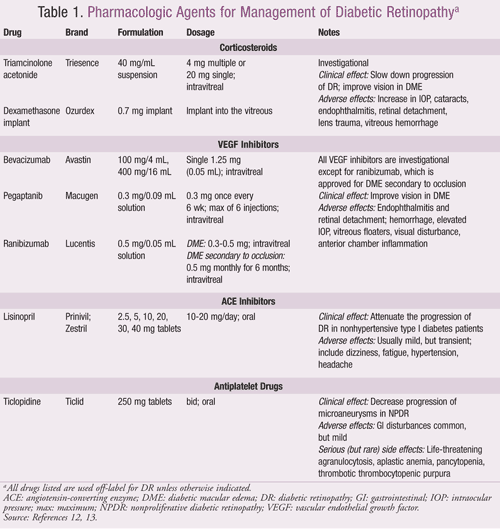
VEGF Inhibitors: VEGF is a suitable therapeutic target because it is upregulated in DR and contributes to neovascularization and capillary leakage in severe DR. Three drugs are currently available in the market—bevacizumab, pegaptanib, and ranibizumab (TABLE 1). While ranibizumab has been approved by the FDA for secondary DME resulting from thrombosis and retinal vein occlusion, all the VEGF inhibitors have found off-label application in the treatment of DME.12 Because VEGF inhibitors are administered intravitreally, adverse systemic effects are minimized. Safety studies are still in progress for this class of drugs.
Renin-Angiotensin System (RAS) Inhibitors: Angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARBs) reduce DR and VEGF levels in PDR.4 Lisinopril, an ACE inhibitor, is used off-label to attenuate DR progression in nonhypertensive type I patients (TABLE 1).13 Therapy may be discontinued if hypotension and hyperkalemia persist. The Diabetic REtinopathy Candesartan Trials (DIRECT) study demonstrated that candesartan, an ARB, decreased the incidence, but not the progression, of DR in normotensive type 1 diabetes patients.14
Antiplatelet Drugs: Platelet aggregation contributes to capillary occlusion and microaneurysm formation in DR. The antiplatelet drug ticlopidine has an off-label indication to delay the rate of microaneurysm formation in NPDR (TABLE 1).13
Protein Kinase C (PKC) Inhibitors: The beta isoform of PKC modulates and upregulates the expression of VEGF, which, in turn, contributes to microvascular DR complications. Ruboxistaurin, a PKC-beta inhibitor, is undergoing evaluation for its ability to prevent end-organ complications in DR. Ongoing clinical trials indicate diarrhea may be a clinically relevant adverse effect.15
Advanced Glycation End-Product (AGE) Inhibitors: Accumulation of AGEs in the diabetic retina appears to contribute to the end-organ damage observed in DR. Pimagedine, also known as aminoguanidine, is an oral AGE inhibitor under investigation for its ability to prevent end-organ complications. Adverse reactions and oral dosing have not been fully elucidated.13
Adherence to Therapy
Patients with diabetes must adhere to prescribed medication regimens for hypertension and hyperglycemia to reduce the risks of DR. Adverse effects, financial concerns, and limited knowledge are problems that may need to be addressed. It is common for patients with diabetes to use multiple drugs. Clear directions, in addition to willingness to comply, are key to effective treatment. The patient must understand his or her regimen and have the proper tools, such as glucometers, sphygmomanometers, insulin pumps, and pill organizers, to improve adherence. Because of vision problems, some persons may need equipment with large displays.
Many insulin-dependent patients use multiple types of insulin, which are categorized by onset and duration of action. It is critical for these patients to know which type of insulin to use when, to avoid the dangers of hypoglycemia and the long-term effects of hyperglycemia, such as DR. The ability to use glucometers and interpret results correctly will assist patients in adhering to the insulin regimen. In addition, patients must comprehend their basal and nutritional needs. Many people fear giving themselves or family members subcutaneous insulin injections. Insulin pumps make drug delivery easier but must be used correctly, and the technology may intimidate some people. Finger sticks can be painful; pharmacists should recommend the least painful monitoring devices available. Because of the many challenges facing individuals with diabetes, it is recommended they participate in diabetes self-management education programs to optimize adherence to medication therapy.6
Conclusion
According to the CDC, nearly 22% of the 4.1 million Americans with DR are at risk for vision-threatening complications.2 Therapies to achieve recommended glycemic and blood pressure targets, and close monitoring of blood glucose and blood pressure, are essential components of a strategy to preserve sight in patients with diabetes. Regular ophthalmic screenings help decrease DR risks by facilitating timely medical interventions.2
REFERENCES
1. Cheung N, Mitchell P, Wong T. Diabetic retinopathy. Lancet. 2010;376:124-136.
2. CDC. Vision Health Initiative. Basic information. Common eye disorders. Diabetic retinopathy. www.cdc.gov/visionhealth/
3. American Academy of Ophthalmology Retina Panel. Preferred Practice Pattern Guidelines. Diabetic Retinopathy. San Francisco, CA: American Academy of Ophthalmology; 2008.
4. Leal EC, Santiago AR, Ambrosio AF. Old and new drug targets in diabetic retinopathy: from biochemical changes to inflammation and neurodegeneration. Cur Drug Targets CNS Neurol Disord. 2005;4:421-434.
5. Mohamed Q, Gillies MC, Wong TY. Management of diabetic retinopathy: a systematic review. JAMA. 2007;298:902-916.
6. American Diabetes Association. Standards of medical care in diabetes–2011. Diabetes Care.
7. Ockrim Z, Yorston D. Managing diabetic retinopathy. Br Med J. 2010;341:930-935.
8. Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329:977-986.
9. The ACCORD Study Group. Effects of medical therapies on retinopathy progression in type 2 diabetes. N Engl J Med. 2010;363:233-244.
10. Schwartz SG, Flynn HW. Pharmacotherapies for diabetic retinopathy: present and future. Exp Diabetes Res. 2007;2007:52487.
11. Herrero-Vanrell R, Cardillo JA, Kuppermann BD. Clinical applications of the sustained-release dexamethasone implant for treatment of macular edema. Clin Ophthalmol. 2011;5:139-146.
12. Nicholson PB, Schachat AP. A review of clinical trials of anti-VEGF agents for diabetic retinopathy. Grafaes Arch Clin Exp Ophthalmol. 2010;248:915-930.
13. Clinical Pharmacology [database online]. www.clinicalpharmacology-ip. 2011;34(suppl 1):S11-S65.
14. Chaturvedl N, Porta M, Klein R, et al; DIRECT Programme Study Group. Effect of candesartan on prevention (DIRECT-Prevent 1) and progression (DIRECT-Protect 1) of retinopathy in type 1 diabetes: randomised, placebo-controlled trials. Lancet. 2008;372:1394-1402.
15. The PKC-DMES Study Group. Effect of ruboxistaurin in patients with diabetic macular edema. Arch Ophthalmol. 2007;125:318-324.
16. Photos, images, and videos. Normal vision; diabetic retinopathy. National Eye Institute, National Institutes of Health. www.nei.nih.gov/photo/keyword.
To comment on this article, contact rdavidson@uspharmacist.com.





