US Pharm.
2008;33(4):36-48.
Raynaud's
phenomenon was named for French physician Maurice Raynaud (1834-1881) and was
first described in 1862. This disorder is referred to as Raynaud's disease
when the cause is unknown and Raynaud's phenomenon when it is
secondary to another condition.1 It is characterized by attacks of
vasospasm of digital arteries and arterioles that leads to intermittent pallor
or cyanosis of the skin. It primarily affects the fingers, usually distal to
the knuckles, and less commonly the toes. More rarely, Raynaudsymptoms may
affect the nose, tongue, and ear lobes. Several case reports have described
Raynaud's as affecting the nipples of breastfeeding women and suggest that
this may be a treatable cause of painful breastfeeding, especially if no other
signs of infection or trauma are present.2-4 These attacks may last
from minutes to hours and are relatively benign in most patients.
Raynaud's phenomenon may be
secondary to many more serious conditions and may foretell the development of
these diseases by months or years. Therefore, any patient who presents with
complaints of Raynaud's symptoms should be carefully evaluated for any
underlying cause, undergo a thorough drug history, and be educated as to the
importance of lifestyle changes that might control disease symptoms.
Epidemiology and
Pathophysiology
Due to diagnostic
uncertainties, the precise incidence of Raynaud's symptoms in the general
population is unclear. Although some sources cite an incidence as high as 20%,
1,5 more recent reviews place the prevalence at 3% to 5%.6 It
most commonly occurs in women of childbearing age.5 Raynaud's
disease, also known as primary Raynaud's, is caused by reversible spasm
of the smallest arteries and arterioles; no abnormalities of the endothelium
are observed. In these cases, the erythrocyte sedimentation rate (ESR),
antinuclear antibodies (ANA), and nailfold capillaries are usually normal.
7 The arterial vasospasm that characterizes Raynaud's is thought to be
secondary to exaggerated reflex sympathetic vasoconstriction. The threshold
for vasospastic response is lowered by anything that activates sympathetic
outflow or leads to the release of catecholamines, such as cold temperatures
or emotional upset.8 A clinical association with migraine
headaches, variant (Prinzmetal's) angina, and pulmonary hypertension suggests
a common mechanism for vasospasm in multiple arterial beds.9 There
definitely appears to be a familial component to primary Raynaud's; smoking
worsens the frequency and intensity of attacks, and there seems to be a
hormonal component to the condition.6,10,11
Primary Raynaud's usually
results in a milder form of the disease than do most cases of Raynaud's
phenomenon, and many cases do not require pharmacologic treatment. The
condition may progress in approximately 30% of patients, although long-term
complications are rarely severe enough to cause gross tissue loss. The skin of
the digits may become smooth, shiny, and tight with the loss of subcutaneous
tissue (sclerodactyly), small painful ulcers may appear on the tips of the
fingers, and, in very advanced cases, the arterial intima may thicken and
thromboses may form in small arteries.
Raynaud's phenomenon, also
known as secondary Raynaud's, usually begins later in life and includes
a variety of pathophysiologic features including vasospasm, endothelial cell
changes, vessel obstruction, and hemorheologic deformations.5,7
Because secondary Raynaud's is associated not only with vasospasm but also
with fixed blood vessel defects, the ischemia is often much more severe and
more difficult to treat.12,13 The differences between primary and
secondary Raynaud's are detailed in TABLE 1.
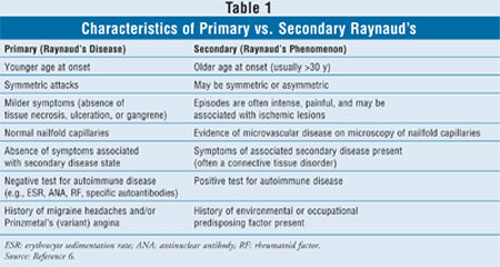
Many serious, potentially
life-threatening conditions may be associated with secondary Raynaud's.
Secondary Raynaud's is most commonly associated with scleroderma (system
sclerosis) or limited scleroderma (CREST syndrome), occurring in 90% to 95% of
all cases.7 More important, secondary Raynaud's may be the initial
manifestation of disease in 70% of patients with scleroderma, and it may
present several years before more obvious signs of connective tissue disease
(CTD) are evident.5 Because the Raynaud's that develops in
conjunction with scleroderma is often very severe and may progress to gangrene
and/or autoamputation, it is important for prognostic and therapeutic reasons
to determine if these symptoms are associated with an underlying condition
that may benefit from earlier and more aggressive treatment.
In a study from 2003, 113
women and 29 men were divided into either "primary" or "possible secondary"
Raynaud's groups. Overall, 20 (14.1%) of these patients progressed to a
definite CTD diagnosis over a median follow-up period of 12.4 years. Factors
associated with transition to a CTD included initial presence of ANA,
thickening of fingers, a higher age at onset of Raynaud's symptoms, and female
sex.14 An earlier meta-analysis involving 639 patients with
presumed primary Raynaud's documented the development of a secondary disorder
in 81 (12.6%) of these patients, at a mean time of 10.4 years from the onset
of Raynaud's symptoms.15
Secondary Raynaud's may also
be associated with several other rheumatologic or dermatologic conditions
including rheumatoid arthritis, systemic lupus erythematosus, dermatomyositis,
and polymyositis. In a review of 105 cases (69 polymyositis, 36
dermatomyositis), it was noted that Raynaud's symptoms were an early
presenting sign in 51% of the cases.16
Atherosclerosis may be
associated with symptoms of secondary Raynaud's, especially in older male
patients. One study assessed 103 patients suffering from Raynaud's phenomenon
without any obvious underlying disease. Forty-four (43%) of these patients
were determined to have atherosclerotic vascular disease, almost one half of
whom had a dyslipidemia, most commonly of familial origin.17 In
younger men who smoke, thromboangiitis obliterans (Buerger's disease) may be
associated with secondary Raynaud's.
Other causes include disorders
that increase blood viscosity (e.g., cryoglobulinemia, myeloproliferative
disorders), cancer, hypothyroidism, anorexia nervosa, neurologic disorders
(e.g., stroke, poliomyelitis, carpal tunnel syndrome), traumatic injuries
(e.g., vibration injury, electric shock injury, cold injury/frostbite),
exposure to vinyl chloride, continued exposure to cold (e.g., frozen food
packing), typing, or piano playing. Secondary Raynaud's is quite common in
people whose jobs involve the use of vibrating equipment (e.g., drills,
jackhammers, and drums).
Secondary Raynaud's may also
be associated with several drug therapies including beta-blockers, ergot
alkaloids, sympathomimetics, clonidine, narcotics, cocaine, methysergide, some
chemotherapeutic agents (e.g., bleomycin, vinblastine, cisplatin), estrogen,
and cyclosporine. Some patients may benefit from a change to a low-estrogen or
progesterone-only oral contraceptive.
Clinical Presentation and
Diagnosis
Raynaud's is
characterized by intermittent attacks of blanching or cyanosis of the digits,
which is usually precipitated by exposure to cold temperatures and/or
emotional upset.6 The affected digits often feel cold and numb, and
there is a typical triphasic series of skin color changes, which proceeds from
pallor (white) to cyanosis (blue) to reactive hyperemia (red). A painful
throbbing may occur during the hyperemic phase, and paresthesias are frequent.
Raynaud's is diagnosed primarily by a history of these classic symptoms, which
are relatable to common precipitating factors, or the finding of a history of
exposure to environmental or occupational factors that are associated with
secondary Raynaud's. No single office or laboratory test confirms the
diagnosis.
In secondary Raynaud's, the
signs and symptoms of the underlying disorder may or may not be present, since
Raynaud's may be an early sign of such disorders. The presence of Raynaud's
symptoms indicates the need to rule out an autoimmune process, which requires
the examination of specific laboratory parameters such as the complete blood
count (CBC), ESR, ANA, rheumatoid factor, and disease-specific autoantibodies.
The finding of distorted capillaries in the nailfolds using an ophthalmoscope
may be the best predictor of associated CTD.6,18 Patients with a
history of single-digit or asymmetric attacks should undergo testing for
presence of large-artery disease, such as vasculitis, atherosclerosis, or
other embolic conditions. Thyroid function tests may be indicated to rule out
hypothyroidism. In older patients who present with new-onset Raynaud's
symptoms, a malignancy should be strongly considered during diagnostic workup,
especially in patients who also present with symptoms consistent with bone
pain. Patients diagnosed with primary Raynaud's should be followed closely for
clinical or laboratory signs that suggest development of a secondary disorder.
If such signs do not develop within a two-year period, some clinicians suggest
that a secondary disease is unlikely.6
Nonpharmacologic Treatment
Most patients with
primary Raynaud's experience only mild and infrequent episodes. Therefore,
patient education, reassurance, and nondrug interventions may often be
adequate.8 Patients should be instructed to avoid inciting
environmental factors such as direct contact with frozen foods or cold drinks.
These patients may need to wear insulation against cold weather or use
electric or chemical warming devices.12 Gloves may need to be worn
in temperatures that would be considered mild by normal individuals. It is
important to remove any predisposing drugs (as previously described) from the
patient's medication regimen. These patients should be strongly encouraged to
stop smoking, which aggravates optimal oxygen delivery to the tissues.12
Biofeedback and relaxation techniques may help some patients, especially
those who demonstrate Raynaud's symptoms secondary to emotional upset. If
ulcerations develop, patients should be encouraged to keep them sterile and to
see a doctor if an infection is present. If such ulcers progress or are
severe, a wound care specialist may need to be consulted.
The first-line therapy for
secondary Raynaud's is to treat the underlying disorder, if possible. These
patients may develop increasingly severe Raynaud's symptoms if the underlying
disorder continues untreated. In addition, as the underlying disease worsens,
the treatment of complications from Raynaud's becomes increasingly difficult.
Pharmacologic Treatment
Several drugs may be used to treat
the symptoms of Raynaud's if nonpharmacologic treatments are inadequate. A
summary of recommended agents is listed in TABLE 2.
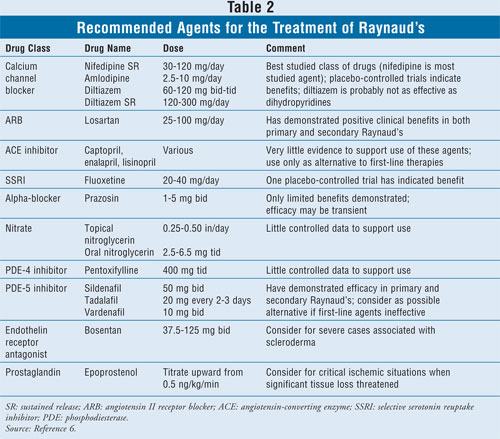
Calcium Channel Blockers:
The most commonly used drugs for the treatment of Raynaud's symptoms are the
dihydropyridine calcium channel blockers (CCBs), of which nifedipine is the
most studied agent. These agents act primarily by increasing vasodilation and
decreasing vasospasm in the affected arteries; however, they may also inhibit
the growth and proliferation of vascular smooth muscle cells, depress T-cell
function, and possess limited antiplatelet activity.19,20 Most of
the early studies demonstrating the efficacy of these agents were done with
immediate-release nifedipine, which led to a significant number of adverse
effects, including headache, hypotension, flushing (~10%), tachycardia, and
peripheral ankle or pedal edema (~30%).12,19
Two recent studies have shed
further light on the efficacy and tolerability of these agents. The Raynaud's
Treatment Study Investigators compared sustained-release (SR) nifedipine with
temperature biofeedback in the treatment of 313 patients with primary
Raynaud's in a randomized trial with one-year follow-up. These patients were
diagnosed by medical history and normal nailfold capillaries and had a history
of two or more attacks per day during the previous cold season. Patients
treated with nifedipine demonstrated a 66% decrease in verified attacks versus
placebo (P <.001); however, adverse effects associated with nifedipine
resulted in therapy discontinuation in 15% of the patients. Temperature
biofeedback training did not decrease attacks significantly versus control
biofeedback. The authors concluded that SR nifedipine is a safe and effective
treatment for primary Raynaud's and is more effective than temperature
biofeedback.1
A meta-analysis investigated
the efficacy of CCBs for the treatment of Raynaud's secondary to scleroderma.
This study analyzed eight randomized trials, which included a total of 109
patients. These patients demonstrated a mean reduction of 8.3 attacks every
two weeks, and the attacks were judged to be about 35% less severe. The
authors concluded that CCBs appear to lead to significant clinical improvement
in both the frequency and severity of ischemic attacks, although the effect
was judged to be "moderate." They also suggested that low dosing may have
contributed to the modest effects.13 However, patients with
secondary disease, in whom structural damage to the vessel wall is pronounced
and fixed, would not be expected to respond as well to CCBs as patients
suffering primarily from vasospasm (primary Raynaud's).
Several other CCBs have also
demonstrated efficacy in the treatment of Raynaud's symptoms, including
amlodipine, felodipine, nisoldipine, and isradipine.21-24
Nicardipine has been investigated in several studies with mixed results.
20,25-27 Although not a dihydropyridine agent, diltiazem has also been
investigated for the treatment of Raynaud's symptoms.28 It is
probably less effective than the dihydropyridine class, and its effectiveness
has been demonstrated for primary Raynaud's only; however, it may be a
reasonable alternative for patients who cannot tolerate the dihydropyridine
agents. The CCBs should be used for up to six weeks before a therapeutic
assessment is made.19
ACE Inhibitors and ARBs:
An excellent review of the use of angiotensin-converting enzyme (ACE)
inhibitors and angiotensin II receptor blockers (ARBs) in the treatment of
Raynaud's was recently published.29 These authors reviewed several
small, short-term studies (12 weeks or less) that had evaluated captopril,
enalapril, and losartan. They concluded that these agents may provide minor
benefits, although no definitive evidence exists to suggest that they are
superior to dihydropyridine CCBs.
However, two of the studies
did find a significantly decreased number of attacks with the use of losartan,
one in patients with primary Raynaud's and one that included patients with
secondary Raynaud's.30,31 The second study directly compared
losartan with nifedi!= pine in a randomized, parallel group, controlled trial
that included 25 patients with primary Raynaud's and 27 patients with
Raynaud's secondary to scleroderma. A reduction in the severity of the
episodes followed both treatments, but the effect was greater in the losartan
arm (P <.05). Episode frequency was decreased only in the losartan
group (P <.01); however, as might be expected, clinical benefit was
greatest in the primary Raynaud's group. The authors suggested that losartan
may have additional disease-modifying potential if used for secondary
Raynaud's associated with scleroderma.31
In conclusion, there is not
convincing evidence to favor these renin-angiotensin mediators over CCBs for
first-line therapy, but they might be recommended as an alternative if CCBs
are ineffective or not tolerated. Losartan may be preferred over the ACE
inhibitors, especially in patients with secondary Raynaud's.
PDE-5 Inhibitors:
The phosphodiesterase type 5 (PDE-5) inhibitors have demonstrated efficacy in
the treatment of Raynaud's symptoms in several case reports and clinical
trials. A comprehensive summary of this topic was recently published.32
These agents (i.e., sildenafil, tadalafil, and vardenafil) decreased the
metabolism of cyclic guanosine monophosphate (cGMP) in vascular smooth muscle
cells, which results in enhanced cGMP-dependent microvascular and
macrovascular dilation. It was originally reported that the treatment of
pulmonary hypertension with sildenafil in a patient with scleroderma was
associated with significant improvement of coexisting secondary Raynaud's.
33 In several subsequent case reports, these agents have been shown to
provide symptomatic improvement, ulcer healing, and objective improvements in
blood flow in patients with both primary and secondary Raynaud's.32
Vardenafil was investigated in
an open-label pilot study of 40 patients, 33 of whom had secondary Raynaud's.
Overall, digital blood flow improved in 70% of these patients, and in the
patients who responded, it improved by 35% during a cold-exposure test after
two weeks of therapy (P <.01). Clinical symptoms improved in 68% of the
patients, and the Raynaud condition score (RCS) improved significantly (P
<.001). The response rates of patients with primary or secondary Raynaud's
were similar, as was the response rates of smokers versus nonsmokers.34
Tadalafil was compared to
pentoxifylline in a small study of patients with severe Raynaud's secondary to
autoimmune disease. The tadalafil-treated patients showed significant
improvement in the RCS and significant decreases in both attack frequency and
duration. These patients also demonstrated continued improvement over the
four-week treatment period.35
Sildenafil was investigated in
a double-blind, placebo-controlled, fixed-dose, crossover study in 16 patients
with symptomatic secondary Raynaud's resistant to vasodilator therapy. While
taking sildenafil, the mean frequency of Raynaud attacks was lower (P =
.0064), the cumulative attack duration was shorter (P = .0038), and the
mean RCS was lower (P= .0386). More impressively, the mean capillary
blood flow velocity for all patients more than quadrupled after treatment with
sildenafil (P = .0004). Only two patients discontinued sildenafil due
to side effects, and 16 patients subsequently requested off-label continuation
of therapy.36 Overall, the PDE-5 inhibitors appear to be a
promising class of agents, especially for the treatment of patients with
severe secondary Raynaud's who have not found relief with first-line
vasodilators.
Fluoxetine:
Fluoxetine, a selective serotonin reuptake inhibitor (SSRI), was compared
with nifedipine for the treatment of Raynaud's symptoms in a study published
in 2003. Serotonin is a selective vasoconstrictor, and infusion into the
brachial artery has resulted in the characteristic sequential color changes
associated with Raynaud's. In addition, anecdotal reports had suggested that
fluoxetine may be beneficial in Raynaud's.37,38 Twenty-six patients
with primary and 27 patients with secondary Raynaud's were randomly assigned
to receive six weeks of treatment with fluoxetine or nifedipine. After a
two-week washout period, each group was crossed over to the other treatment
arm. Results demonstrated a reduction in attack frequency and severity in
patients treated with either agent; however, the effect was statistically
significant only in the fluoxetine-treated group. Subgroup analysis showed
that the greatest response was seen in females and in patients with primary
Raynaud's. No significant adverse effects occurred in the fluoxetine-treated
group.39
Bosentan and Iloprost:
For severe cases of secondary Raynaud's, usually associated with scleroderma,
several studies and case reports have supported the use of either the oral
endothelin receptor antagonist bosentan or the IV administration of the
synthetic prostacyclin (PGI2) analogue iloprost. The efficacy of
bosentan was suggested through its use for pulmonary arterial hypertension in
scleroderma patients who experienced coincident prevention or healing of skin
ulcers associated with severe Raynaud's.40,41 The efficacy of this
agent has been reported primarily via case reports; however, the reported
clinical benefits have been quite impressive in patients with severe tissue
injury.42-45 A placebo-controlled, double-blind, prospective study
of patients with pulmonary hypertension receiving bosentan demonstrated a 48%
reduction in the mean number of new ulcers during the 16-week treatment
period. In addition, a statistically significant improvement in hand function
was observed. There was no difference between treatment groups in healing of
existing ulcers.46
A study reported in 1989
compared the long-term effects of short-term iloprost infusion with those of
nifedi!= pine in patients with Raynaud's secondary to scleroderma. Patients
received iloprost at 2 ng/kg/minute for eight hours on three consecutive days
with a further single infusion at week 8. The nifedipine dose was increased to
60 mg/day during the study. Both regimens produced a decrease in the number,
duration, and severity of attacks; the mean number of digital lesions was also
decreased with both treatments. However, more adverse effects were associated
with nifedipine therapy. This study concluded that both iloprost and
nifedipine are beneficial in the treatment of secondary Raynaud's.47
A more recent trial evaluated
the effects of long-term cyclic therapy with iloprost compared with nifedipine
on skin score and RCS in patients with Raynaud's secondary to scleroderma.
Iloprost significantly reduced the skin score compared to nifedipine; however,
there was no significant difference in RCS scores.48 IV iloprost is
not available in the U.S.; however, a related agent, epoprostenol, might be
considered for patients with critical ischemia.6
Other Agents:
Both oral and topical nitroglycerin products have been used to treat the
symptoms associated with Raynaud's; however, little controlled data exist to
support the use of these agents, and systemic products are associated with
bothersome side effects such as flushing, headache, and hypotension.6,12
The alpha1-receptor blocker prazosin has been found in two
randomized controlled trials to be more effective than placebo in the
treatment of Raynaud's secondary to scleroderma. However, the positive
response is modest, and side effects such as dizziness, hypotension, and
palpitations, may be limiting.49 Pentoxifylline, with or without
aspirin, has been commonly used, but there is very little clinical trial
information supporting its use. These therapies may be best reserved for more
severely affected patients who have evidence of endothelial damage. These two
agents, in combination with nifedi!= pine, may be effective in patients
suffering from hand-arm vibration syndrome.50 The selective PDE-3
inhibitor cilostazol, which inhibits platelet aggregation and acts as a
vasodilator, was studied in 40 subjects with Raynaud's. This agent increased
brachial artery diameter but did not demonstrate positive benefits regarding
microvascular flow or disease symptoms.51
Summary
Raynaud's is a
disorder classically associated with transient digital ischemia in response to
cold or emotional stress. It is classified into two distinct entities, which
suggests important differences in diagnostic strategies, prognoses, and
therapeutic choices. Primary Raynaud's (Raynaud's disease) is idiopathic,
symmetric, associated with normal nailfold capillaries, generally presents an
uncomplicated, self-limited course, and often responds adequately to
nonpharmacologic measures. Secondary Raynaud's (Raynaud's phenomenon) is
usually associated with an older age at onset, abnormal vessel pathology, the
presence of a secondary cause on the basis of history and/or physical
examination, a more severe clinical course (e.g., tissue necrosis,
ulcerations, gangrene), and the need for definitive pharmacologic therapy
Important nonpharmacologic
treatments include cold avoidance, stress management, and smoking cessation.
When pharmacologic therapy is indicated, a dihydro!= pyr!= i!= dine CCB is
usually preferred as first-line therapy. Rational alternatives include
diltiazem, losartan, fluoxetine, or possibly an ACE inhibitor. For more severe
cases, or those not responsive to these therapies, a PDE-5 inhibitor,
bosentan, or, in very severe cases, epoprostenol may be considered. For
patients with fixed arterial pathology, pentoxifylline and/or low-dose aspirin
may be beneficial. In addition, in secondary Raynaud's, it is critical to
treat the underlying cause as effectively as possible.
REFERENCES
1. Raynaud
Treatment Study Investigators. Comparison of sustained-release nifedipine and
temperature biofeedback for treatment of primary Raynaud phenomenon. Arch
Intern Med. 2000;160:1101-1108.
2. Page SM, McKenna DS.
Vasospasm of the nipple presenting as painful lactation. Obstet Gynecol.
2006;108:806-808.
3. Anderson JE, Held N,
Wright K. Raynaud's phenomenon of the nipple: a treatable cause of painful
breastfeeding. Pediatrics. 2004;113:360-364.
4. Garrison CP. Nipple
vasospasms, Raynaud's syndrome, and nifedipine. J Hum Lact.
2002;18:382-385.
5. Bolster MB, Maricq
HR, Leff RL. Office evaluation and treatment of Raynaud's phenomenon. Cleve
Clin J Med. 1995;62:51-61.
6. Wigley FM. Raynaud's
phenomenon. N Engl J Med. 2002;347:1001-1008.
7. Lambova SN,
Kuzmanova SI. Raynaud's phenomenon in common rheumatic diseases. Folia Med.
2006;48:22-28.
8. Brown KM, Middaugh
SJ, Haythornthwaite JA, Bielory L. The effects of stress, anxiety, and outdoor
temperature on the frequency and severity of Raynaud's attacks: the Raynaud's
Treatment Study. J Behav Med. 2001;24:137-153.
9. O'Keeffe ST,
Tsapatsaris NP, Beetham WP. Increased prevalence of migraine and chest pain in
patients with primary Raynaud disease. Ann Intern Med.
1992;116:985-989.
10. Turton EP, Kent PJ,
Kester RC. The aetiology of Raynaud's phenomenon. Cardiovasc Surg.
1998;6:431-440.
11. Fraenkel L, Yuqing
Z, Chaisson CE, et al. The association of estrogen replacement therapy and
Raynaud phenomenon in postmenopausal women. Ann Intern Med.
1998;129:208-211.
12. Pope JE. The
diagnosis and treatment of Raynaud's phenomenon: a practical approach.
Drugs. 2007;67:517-525.
13. Thompson AE, Shea
B, Welch V, et al. Calcium-channel blockers for Raynaud's phenomenon in
systemic sclerosis. Arthritis Rheum.2001;44:1841-1847.
14. Ziegler S, Brunner
M, Eigenbauer E, Minar E. Long-term outcome of primary Raynaud's phenomenon
and its conversion to connective tissue disease: a 12-year retrospective
patient analysis. Scand J Rheumatol. 2003;32:343-347.
15. Spencer-Green G.
Outcomes in primary Raynaud phenomenon: a meta-analysis of the frequency,
rates, and predictors of transition to secondary diseases. Arch Intern Med.
1998;158:595-600.
16. Tymms KE, Webb J.
Dermatopolymyositis and other connective tissue diseases: a review of 105
cases. J Rheumatol. 1985;12:1140-1148.
17. van Vugt RM, Kater
L, Dijkstra PF, et al. The outcome of angiography in patients with Raynaud's
phenomenon: an unexpected role for atherosclerosis and hypercholesterolemia.
Clin Exp Rheumatol. 2003;21:445-450.
18. Nagy Z, Czirjak L.
Nailfold digital capillaroscopy in 447 patients with connective tissue disease
and Raynaud's disease. Eur Acad Dermatol Venereol. 2004;18:62-68.
19. Palamaras I,
Kyriakis K. Calcium antagonists in dermatology: a review of the evidence and
research-based studies. Dermatology Online J. 2005;11:8.
20. Wigley FM, Wise RA,
Malamet R, Scott TE. Nicardipine in the treatment of Raynaud's phenomenon.
Dissociation of platelet activation from vasospasm. Arthritis Rheum.
1987;30:281-286.
21. La Civita L, Pitaro
N, Rossi M, et al. Amlodipine in the treatment of Raynaud's phenomenon. Br
J Rheumatol. 1993;32:524-525.
22. Kallenberg CG,
Wouda AA, Meems L, Wesseling H. Once daily felodipine in patients with primary
Raynaud's phenomenon. Eur J Clin Pharmacol. 1991;40:313-315.
23. Gjorup T, Hartling
OJ, Kelbaek H, Nielsen SL. Controlled double-blind trial of nisoldipine in the
treatment of idiopathic Raynaud's phenomenon. Eur J Clin Pharmacol.
1986;31:387-389.
24. Leppert J, Jonasson
T, Nilsson H, Ringqvist I. The effect of isradipine, a new calcium-channel
antagonist, in patients with primary Raynaud's phenomenon: a single-blind dose
response study. Cardiovasc Drugs Ther. 1989;3:397-401.
25. Wollersheim H,
Thien T. Double-blind placebo-controlled crossover study of oral nicardipine
in the treatment of Raynaud's phenomenon. J Cardiovasc Pharmacol.
1991;18:813-818.
26. Ferri C, Cecchetti
R, Cini G, et al. Slow-releasing nicardipine in the treatment of Raynaud's
phenomena without underlying diseases. Clin Rheumatol. 1992;11:76-80.
27. Kahan A, Amor B,
Menkes CJ, et al. Nicardipine in the treatment of Raynaud's phenomenon: a
randomized double-blind trial. Angiology. 1987;38:333-337.
28. Kahan A, Amor B,
Menkes CJ. A randomized double-blind trial of diltiazem in the treatment of
Raynaud's phenomenon. Ann Rheum Dis. 1985;44:30-33.
29. Wood HM, Ernst ME.
Renin-angiotensin system mediators and Raynaud's phenomenon. Ann
Pharmacother. 2006;40:1998-2002.
30. Pancera P, Sansone
S, Secchi S, et al. The effects of thromboxane A2 inhibition
(picotamide) and angiotensin II receptor blockade (losartan) in primary
Raynaud's phenomenon. J Intern Med. 1997;242:373-376.
31. Dziadzio M, Denton
CP, Smith R, et al. Losartan therapy for Raynaud's phenomenon and scleroderma:
clinical and biochemical findings in a fifteen-week, randomized,
parallel-group, controlled trial. Arthritis Rheum. 1999;42:2646-2655.
32. Levien TL.
Phosphodiesterase inhibitors in Raynaud's phenomenon. Ann Pharmacother.
2006;40:1388-1393.
33. Rosenkranz S, Diet
F, Karasch T, et al. Sildenafil improves pulmonary hypertension and peripheral
blood flow in a patient with scleroderma-associated lung fibrosis and
Raynaud's phenomenon. Ann Intern Med. 2003;139:871-873.
34. Caglayan E,
Huntgeburth M, Karasch T, et al. Phosphodiesterase type 5 inhibition is a
novel therapeutic option in Raynaud disease. Arch Intern Med.
2006;166:231-233.
35. Carlino G.
Treatment of Raynaud's phenomenon with tadalafil, a phosphodiesterase-5
inhibitor [abstract]. Ann Rheum Dis. 2005;64(suppl 3):258.
36. Fries R, Sharlat K,
von Wilmowsky H, Bohm M. Sildenafil in the treatment of Raynaud's phenomenon
resistant to vasodilator therapy. Circulation. 2005;112:2980-2985.
37. Jaffe IA. Serotonin
reuptake inhibitors in Raynaud's phenomenon. Lancet. 1995;345:1378.
38. Bolte MA, Avery D.
Case of fluoxetine-induced remission of Raynaud's phenomenon--a case report.
Angiology. 1993;44:161-163.
39. Coleiro B, Marshall
SE, Denton CP, et al. Treatment of Raynaud's phenomenon with the selective
serotonin reuptake inhibitor fluoxetine. Rheumatology (Oxford).
2001;40:1038-1043.
40. Black CM, Korn JH,
Mayes MD, Matucci-Cerinic M. Improvements in the net ulcer burden and hand
functionality in patients with digital ulcers related to systemic sclerosis.
Arthritis Rheum.2003;47(suppl):1137.
41. Humbert M, Cabane
J. Successful treatment of systemic sclerosis digital ulcers and pulmonary
arterial hypertension with endothelin receptor antagonist bosentan.
Rheumatology (Oxford). 2003;42:191-193.
42. Ramos-Casales M,
Brito-Zeron P, Nardi N, et al. Successful treatment of severe Raynaud's
phenomenon with bosentan in four patients with systemic sclerosis.
Rheumatology (Oxford). 2004;43:1454-1456.
43. Snyder MJ, Jacobs
MR, Grau RG, et al. Resolution of severe digital ulceration during a course of
bosentan therapy. Ann Intern Med. 2005;142:802-803.
44. Dunne J, Dutz J,
Shojania K, et al. Treatment of severe Raynaud's phenomenon with bosentan in a
patient with systemic sclerosis. Rheumatology (Oxford).
2006;45:911-912.
45. Chamaillard M,
Heliot-Hosten I, Constans J, Taieb A. Bosentan as rescue therapy in
scleroderma refractory digital ulcers. Arch Dermatol. 2007;143:125-126.
46. Korn JH, Mayes M,
Matucci-Cerinic M, et al. Digital ulcers in systemic sclerosis: prevention by
treatment with bosentan, an oral endothelin receptor antagonist. Arthritis
Rheum. 2004;50:3985-3993.
47. Rademaker M, Cooke
ED, Almond NE, et al. Comparison of intravenous infusions of iloprost and oral
nifedipine in treatment of Raynaud's phenomenon in patients with systemic
sclerosis: a double blind randomized study. BMJ. 1989;298:561-564.
48. Scorza R, Caronni
M, Mascagni B, et al. Effects of long-term cyclic iloprost therapy in systemic
sclerosis with Raynaud's phenomenon. A randomized, controlled study. Clin
Exp Rheumatol. 2001;19:503-508.
49. Pope J, Fenlon D,
Thompson A, et al. Prazosin for Raynaud's phenomenon in progressive systemic
sclerosis. Cochrane Database Syst Rev. 2000;(2):CD000956.
50. Buell C, Tobinick
E, Lamp K. Resolution of chronic pain and fingertip ulceration due to hand-arm
vibration syndrome following combination pharmacotherapy. Arch Dermatol.
2007;143:1343-1344.
51. Rajagopalan S,
Pfenninger D, Somers E, et al. Effects of cilostazol in patients with
Raynaud's syndrome. Am J Cardiol. 2003;92:1310-1315.
To comment on this article,
contact rdavidson@jobson.com.





